UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the year ended
OR
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission File Number
(Exact name of registrant as specified in its charter) |
| ||
(State or other jurisdiction of incorporation or organization) |
| (I.R.S. Employer Identification No.) |
|
|
|
| ||
| ||
(Address of principal executive offices) |
| (Zip code) |
(
(Registrant’s telephone number, including area code)
Not Applicable
(Former name, former address and former fiscal year, if changed since last report)
Securities registered pursuant to Section 12(b) of the Act:
|
|
|
| Name of exchange |
Title of each class |
| Trading Symbol |
| on which registered: |
|
| The |
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer | ☐ | Accelerated filer | ☐ |
☒ | Smaller reporting company | ||
|
| Emerging growth company |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act.
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report.
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes
The aggregate market value of the voting stock held by non-affiliates of the registrant as of April 8, 2024 was approximately $
There were
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrant’s definitive proxy statement for its 2024 Annual Meeting of Stockholders (the “Proxy Statement”), to be filed within 120 days of the registrant’s fiscal year ended December 31, 2023, are incorporated by reference in Part III of this Annual Report on Form 10-K. Except with respect to information specifically incorporated by reference in this Annual Report on Form 10-K, the Proxy Statement is not deemed to be filed as part of this Annual Report on Form 10-K.
ASP Isotopes Inc.
Annual Report on Form 10-K
For the Year Ended December 31, 2023
Table of Contents
| 2 |
| Table of Contents |
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K contains forward-looking statements within the meaning of the federal securities laws. All statements other than statements of historical fact contained in this Annual Report on Form 10-K, including statements regarding our future results of operations and financial position, business strategy and plans and objectives of management for future operations, are forward-looking statements. These statements involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements.
In some cases, you can identify forward-looking statements by terms such as “may,” “should,” “would,” “expects,” “plans,” “anticipates,” “could,” “intends,” “target,” “projects,” “contemplates,” “believes,” “estimates,” “predicts,” “potential” or “continue” or the negative of these terms or other similar expressions. The forward-looking statements in this Annual Report on Form 10-K are only predictions. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our business, financial condition and results of operations. These forward-looking statements speak only as of the date of this Annual Report on Form 10-K and are subject to a number of risks, uncertainties and assumptions described in the section titled “Risk Factors” and elsewhere in this Annual Report on Form 10-K. Because forward-looking statements are inherently subject to risks and uncertainties, some of which cannot be predicted or quantified, you should not rely on these forward-looking statements as predictions of future events. The events and circumstances reflected in our forward-looking statements may not be achieved or occur and actual results could differ materially from those projected in the forward-looking statements. Some of the key factors that could cause actual results to differ from our expectations include:
· | our ability to complete the construction of, commission and successfully operate isotope enrichment plants in a cost-effective manner; |
|
|
· | our ability to meet, and to continue to meet, applicable regulatory requirements for the use of the isotopes we may produce using the ASP technology or the Quantum Enrichment process; |
|
|
· | our ability to obtain regulatory approvals for the production and distribution of isotopes; |
|
|
· | our ability to comply on an ongoing basis with the numerous regulatory requirements applicable to the ASP technology, the Quantum Enrichment process and our enrichment facilities in South Africa; |
|
|
· | the introduction, market acceptance and success of Mo-100 that we may produce using ASP technology as an alternative and potentially more convenient production route for Tc-99m; |
|
|
· | the success or profitability of our future offtake arrangements with respect to various isotopes that we may produce using ASP technology or the Quantum Enrichment process; |
|
|
· | a failure of demand for various isotopes that we may produce using ASP technology or the Quantum Enrichment process; |
|
|
· | our future capital requirements and sources and uses of cash; |
|
|
· | our ability to obtain funding for our operations and future growth; |
|
|
· | the extensive costs, time and uncertainty associated with new technology development; |
|
|
· | developments and projections relating to our competitors and industry; |
|
|
· | the ability to recognize the anticipated benefits of acquisitions, including our acquisition of assets of Molybdos (Pty) Limited in the “business rescue” auction, the assets and intellectual property we acquired from Klydon Proprietary Ltd, and our investment in PET Labs Pharmaceuticals; |
|
|
· | problems with the performance of the ASP technology or the Quantum Enrichment process in the enrichment of isotopes; |
|
|
· | our dependence on a limited number of third-party suppliers for certain components; |
|
|
· | our inability to adapt to changing technology and diagnostic landscape, such as the emergence of new diagnostic scanners or tracers; |
|
|
· | our expected dependence on a limited number of key customers for isotopes that we may produce using ASP technology or the Quantum Enrichment process; |
|
|
· | our inability to protect our intellectual property and the risk of claims that we have infringed on the intellectual property of others; |
|
|
· | our inability to compete effectively; |
|
|
· | risks associated with the current economic environment; |
|
|
· | risks associated with our international operations; |
|
|
· | we are subject to credit counterparty risks; |
|
|
· | geopolitical risk and changes in applicable laws or regulations; |
|
|
· | our inability to adequately protect our technology infrastructure; |
|
|
· | our inability to hire or retain skilled employees and the loss of any of our key personnel; |
|
|
· | operational risk; |
|
|
· | costs and other risks associated with becoming a reporting company and becoming subject to the Sarbanes-Oxley Act; |
|
|
· | our inability to implement and maintain effective internal controls; and |
|
|
· | other factors that are described in “Risk Factors,” beginning on page 23. |
| 3 |
| Table of Contents |
These statements relate to future events or to our future financial performance and involve known and unknown risks, uncertainties, and other factors that may cause our actual results, performance, or achievements to be materially different from any future results, performance, or achievements expressed or implied by these forward-looking statements. Factors that may cause actual results to differ materially from current expectations include, among other things, those set forth in Part I, Item 1A - “Risk Factors” below and for the reasons described elsewhere in this Annual Report on Form 10-K. Any forward-looking statement in this Annual Report on Form 10-K reflects our current view with respect to future events and is subject to these and other risks, uncertainties, and assumptions relating to our operations, results of operations, industry, and future growth. Given these uncertainties, you should not place undue reliance on these forward-looking statements. Except as required by law, we assume no obligation to update or revise these forward-looking statements for any reason, even if new information becomes available in the future.
This Annual Report on Form 10-K also contains estimates, projections, and other information concerning our industry, our business, and the potential markets for certain isotopes, including data regarding the estimated size of those markets, their projected growth rates, and the incidence of certain medical conditions. Information that is based on estimates, forecasts, projections, or similar methodologies is inherently subject to uncertainties, and actual events or circumstances may differ materially from events and circumstances reflected in this information. Unless otherwise expressly stated, we obtained these industry, business, market, and other data from reports, research surveys, studies, and similar data prepared by third parties, industry, medical and general publications, government data, and similar sources. In some cases, we do not expressly refer to the sources from which these data are derived.
Except where the context otherwise requires, in this Annual Report on Form 10-K, “we,” “us,” “our,” “ASP Isotopes,” and the “Company” refer to ASP Isotopes Inc. and, where appropriate, its consolidated subsidiaries.
Trademarks
All trademarks, service marks, and trade names included in this Annual Report on Form 10-K are the property of their respective owners. Solely for convenience, the trademarks and trade names in this report may be referred to without the ® and ™ symbols, but such references should not be construed as any indicator that their respective owners will not assert, to the fullest extent under applicable law, their rights thereto.
PART I
Item 1. Business
Overview
We are a development stage advanced materials company dedicated to the development of technology and processes that, if successful, will allow for the enrichment of natural isotopes into higher concentration products, which could be used in several industries. Our proprietary technology, the Aerodynamic Separation Process (“ASP technology”), originally developed by Klydon Proprietary Ltd (“Klydon”), is designed to enable the production of isotopes used in several industries. Our initial focus is on the production and commercialization of enriched Carbon-14 (“C-14”), Molybdenum-100 (“Mo-100”) and Silicon-28 (“Si-28”). We have commissioned an isotope enrichment plant for the enrichment of C-14 located in Pretoria, South Africa, which will be ready for production upon the final installation of essential components. We anticipate completion and commissioning of a multi-isotope enrichment plant in Pretoria, South Africa in mid-2024. In addition, we have started planning additional isotope enrichment plants. We believe the C-14 we may produce using the ASP technology could be used in the development of new pharmaceuticals and agrochemicals. We believe the Mo-100 we may produce using the ASP technology could have significant potential advantages for use in the preparation of nuclear imaging agents by radiopharmacies and others in the medical industry. We believe the Si-28 we may produce using the ASP technology may be used to create advanced semiconductors and in quantum computing. In addition, we are considering the future development of the ASP technology for the separation of Zinc-68, Xenon-129/136 for potential use in the healthcare end market, Germanium 70/72/74 for possible use in the semiconductor end market, and Chlorine -37 for potential use in the nuclear energy end market.
We are also developing Quantum Enrichment technology to produce enriched Ytterbium-176, Nickel-64, Lithium 6, Lithium7 and Uranium-235 (“U-235”). Quantum enrichment is an advanced isotope enrichment technique that is currently in development that uses lasers. We believe that the U-235 we may produce using quantum enrichment technology may be commercialized as a nuclear fuel component for use in the new generation of high-assay low-enriched uranium (HALEU)-fueled small modular reactors that are now under development for commercial and government uses.
| 4 |
| Table of Contents |
The aerodynamic separation technique has its origins in the South African uranium enrichment program in the 1980s, and the ASP technology has been developed during the last 18 years by the scientists at Klydon. In Klydon’s testing, the ASP technology has demonstrated efficacy and commercial scalability in enriching oxygen-18 and silicon-28. ASP Isotopes Inc. was incorporated in Delaware in September 2021 to acquire assets and license intellectual property rights related to the production of Mo-100 using the ASP technology. In January 2022, we also licensed intellectual property rights associated with the production of U-235 using the ASP technology. In July 2022, we licensed intellectual property rights related to the production of all isotopes using the ASP technology. In April 2023, we acquired certain intellectual property assets of Klydon.
We operate principally through subsidiaries: ASP Isotopes Guernsey Limited (the holding company of ASP Isotopes South Africa (Proprietary) Limited and Enlightened Isotopes (Pty) Ltd), which will be focused on the development and commercialization of high-value, low-volume isotopes for highly specialized end markets (such as C-14, Mo-100, and Si-28). In September 2023, we formed a new subsidiary, Quantum Leap Energy LLC, which also has a subsidiary in the United Kingdom (Quantum Leap Energy Ltd), to focus on the development and commercialization of advanced nuclear fuels such as HALEU and Lithium-6. ASP Isotopes UK Ltd is the owner of our technology. In addition, in the fourth quarter of 2023, we entered into a strategic relationship with Pet Labs Pharmaceuticals Proprietary Limited (PET Labs) by acquiring a 51% ownership stake in PET Labs. We anticipate this transaction will allow us to enter the downstream medical isotope production and distribution market.
Our corporate structure and ownership of our subsidiaries is set forth in the chart below:

Recent Events and Key Milestones
Offering of Convertible Notes of Quantum Leap Energy LLC
On February 29, 2024, our wholly owned subsidiary, Quantum Leap Energy LLC (“QLE”), entered into a Convertible Note Purchase Agreement (the “Purchase Agreement”) with certain institutional and individual investors (collectively, the “Purchasers”), to issue and sell to the Purchasers convertible promissory notes of QLE (the “QLE Notes”) in an offering to non-U.S. persons outside of the United States under Regulation S of the Securities Act of 1933, as amended (the “Securities Act”). The closing of the offering of QLE Notes occurred on March 7, 2024 and resulted in gross proceeds to QLE of approximately $20.5 million. We intend to use the net proceeds from the QLE Notes offering for planning, development and construction of QLE’s laser enrichment production facilities and for other general corporate purposes.
We engaged Ocean Wall Limited (the “Placement Agent”) to act as QLE’s sole placement agent in connection with the offering of QLE Notes, pursuant to a placement agency agreement (the “Placement Agent Agreement”), dated as of February 29, 2024, between the Company, QLE and the Placement Agent. Pursuant to the Placement Agent Agreement, QLE agreed to pay the Placement Agent a fee equal to 5.0% of the gross proceeds received by QLE from the sale of QLE Notes, which was paid 50% in cash and 50% in the form of a convertible promissory note in substantially the same form and with substantially the same terms as the QLE Notes.
| 5 |
| Table of Contents |
In connection with the offering of QLE Notes, on February 29, 2024, QLE and the Purchasers entered into a registration rights agreement (the “Registration Rights Agreement”). Under the Registration Rights Agreement, all units or shares of QLE common equity issuable upon conversion of the QLE Notes will be deemed “Registrable Securities.” Under the Registration Rights Agreement holders of the QLE Notes have been granted certain long-form and short-form demand registration rights with respect to the Registrable Securities, including the right to demand an initial public offering (IPO) if QLE has not gone public within five years of the date of the agreement. In addition, holders of the QLE Notes have been granted piggyback registration rights with respect to the Registrable Securities. Certain cash penalties will apply to QLE in the event of registration failures, as described in the Registration Rights Agreement.
Intercompany Agreements between ASP Isotopes Inc. and Quantum Leap Energy LLC
In anticipation of the closing of the offering of QLE Notes, the Company: (1) caused ASP Isotopes UK Limited to enter into a License Agreement, dated as of February 16, 2024, among ASP Isotopes UK Limited, as licensor, and QLE and Quantum Leap Energy Limited (QLE’s UK subsidiary), as licensee, pursuant to which, among other things, the licensee has licensed from the Company the rights to technologies and methods used to separate Uranium-235 and Lithium-6 (including but not limited to the quantum enrichment and ASP technologies) in exchange for a royalty payment in the amount of 10% of QLE revenues (the “License Agreement”); (2) entered into an EPC Services Framework Agreement, dated as of February 16, 2024, with QLE, pursuant to which, among other things, the Company has agreed to provide services for the engineering, procurement and construction of one or more turnkey Uranium-235 and Lithium-6 enrichment facilities in locations to be identified by QLE and owned or leased by QLE, and to commission, start-up and test each such facility, in each case subject to the receipt of all applicable regulatory approvals, permits, licenses, authorizations, registrations, certificates, consents, orders, variances and similar rights (the “EPC Services Agreement”); and (3) effective as of February 16, 2024, assigned to QLE certain existing memoranda of understandings between the Company and certain small modular reactor companies.
Share Purchase Agreement relating to PET Labs
On October 31, 2023, we entered into a Share Purchase Agreement with Nucleonics Imaging Proprietary Limited, a company incorporated in the Republic of South Africa (the “Seller”), relating to the purchase and sale of ordinary shares in the issued share capital of Pet Labs Pharmaceuticals Proprietary Limited, a company incorporated in the Republic of South Africa (“PET Labs”). PET Labs is a South African radiopharmaceutical operations company, dedicated to nuclear medicine and the science of radiopharmaceutical production.
Under the Purchase Agreement, we have agreed to purchase from the Seller 51 ordinary shares in the issued share capital of PET Labs (the “Initial Sale Shares”) (representing 51% of the issued share capital of PET Labs) and we have an option to purchase from the Seller the remaining 49 ordinary shares in the issued share capital of PET Labs (the “Option Shares”) (representing the remaining 49% of the issued share capital of PET Labs). We agreed to pay to the Seller an aggregate of $2,000,000 for the Initial Sale Shares, of which aggregate amount $500,000 was paid on the completion of the sale of the Initial Sale Shares and $1,500,000 is payable on demand after one calendar year from the agreement date. If we exercise our option to purchase the Option Shares (which option is exercisable from the agreement date until January 31, 2027, provided that the Initial Sale Shares have been paid for in full), we have agreed to pay an additional $2,200,000 for the Option Shares.
Company-Owned ASP Plants
In October 2021 and July 2022, we acquired two incomplete ASP plants in Pretoria, South Africa. In September 2021, we obtained the required licenses from the nuclear regulators, including the Non-proliferation Council of South Africa, to complete construction of the plants. We have commissioned the isotope enrichment plant for the enrichment of C-14, which will be ready for production upon the final installation of essential components. We anticipate completion and commissioning of a multi-isotope enrichment plant in Pretoria, South Africa in mid-2024. As of December 31, 2023, we employed 47 persons at this facility.
| 6 |
| Table of Contents |
Supply Contracts
In July 2023, we entered into a supply agreement with a U.S. customer to supply a highly enriched metal. This contract has an annual sales value of $9 million, which we expect to realize during 2024. In September 2023, we received a prepayment of approximately $900,000 for this contract.
In July 2023, we entered into a Memorandum of Understanding (MOU) with a U.S. Small Modular Reactor company to supply HALEU. The MOU focused on formalizing a collaboration to develop a HALEU production facility with financial support from the customer.
In June 2023, we entered into a multi-year supply agreement with a Canadian Customer for the supply of Carbon-14, which will be produced from our facility that was completed in March 2023. The customer will supply carbon-14 in the form of carbon-dioxide gas. We will then convert the carbon dioxide gas into methane under a chemical converting contract entered in June 2023. We will then enrich the methane to greater than 85% C-14 under a tolling agreement, also entered in June 2023. Finally, we will convert the enriched methane back into enriched carbon dioxide under a chemical converting contract. The tolling agreement has a minimum “take or pay” amount of approximately $2.5 million per year, supported by a bank letter of guarantee. In September 2023, we entered into a Memorandum of Understanding (MOU) with the same customer to separate Deuterium and Tritium currently stored at nuclear sites within Canada. The timing and commercial implications of this MOU are subject to future agreement between the parties.
In November 2022, our wholly owned subsidiary, ASP Isotopes Guernsey Limited (“ASP Guernsey”), entered into an Amended Agreement with Klydon (Proprietary) Ltd (“Klydon”) related to a 25-year supply agreement for up to $27 million per annum of highly enriched Molybdenum-100 entered into by and between Klydon and Beijing BRICEM Science and Technology Co. Ltd. (“Bricem”) in August 2021 (the “August 2021 Purchase Agreement”). Under the Amended Agreement, Klydon assigned all of its rights under the August 2021 Purchase Agreement to ASP Guernsey.
Our Segments
As of December 31, 2023, we managed our operations as a single segment, specialist isotopes and related services. Beginning in 2024, primarily as a result of the increased business activities of our subsidiary, Quantum Leap Energy LLC, we will have two operating segments: (i) nuclear fuels, and (ii) specialist isotopes and related services:
| · | Nuclear Fuels. This segment is focused on research and development of technologies and methods used to produce high-assay low-enriched uranium (HALEU) and Lithium-6 for the advanced nuclear fuels target end market. |
|
|
|
| · | Specialist Isotopes and Related Services. This segment is focused on research and development of technologies and methods used to separate high-value, low-volume isotopes (such as C-14, Mo-100 and Si-28) for highly specialized target end markets other than advanced nuclear fuels, including pharmaceuticals and agrochemicals, nuclear medical imaging and semiconductors, as well as services related to these isotopes, and this segment includes PET Labs. |
Our Strategy
Complete development and commissioning of our enrichment facilities in Pretoria, South Africa.
We intend to complete the development and construction of our second enrichment facility located in Pretoria, South Africa, in mid-2024. Our first facility, which will be ready for production upon the final installation of essential components, is designed to enrich light isotopes such as Carbon-14. The second facility, which is substantially larger than the first, should have the potential to enrich kilogram quantities of relatively heavier isotopes, including but not limited to Molybdenum-100 and Silicon-28.
In October 2021, we acquired physical assets, including equipment, of Molybdos (Pty) Limited (Molybdos) located at the plant after having been declared the winner of a competitive auction process under Section 45 of the South Africa Consumer Protection Act, 2008 (the Molybdos Business Rescue Auction). We licensed the ASP technology for the production of Mo-100 from Klydon. We subsequently entered into a turnkey contract with Klydon, pursuant to which Klydon agreed to provide us with a first commercial-scale isotope enrichment plant. The activities to be undertaken or performed by Klydon included taking control of the assets acquired by us in the Molybdos Business Rescue Auction; the design of an enrichment facility; the supply of required components, equipment, and labor; the installation, testing, and commissioning of the enrichment facility; securing all required approvals, regulatory authorizations and other required consents for the operation of the plant; providing training to local ASP Isotopes South Africa (Proprietary) Limited personnel to enable them to operate the plant going forward; and providing warranties in relation to the performance targets of the plant which are required to be met. Klydon was also responsible for liaising with the relevant South African authorities, including the South African Non-Proliferation Council, the Nuclear Suppliers Group, and International Atomic Energy Agency, to ensure that the enrichment plant complied with international laws and guidelines.
In July 2022, we acquired a pilot plant previously used by Klydon to enrich Silicon-28 up to an abundance of 96.6%. This enriched Silicon-28 was then used for experimental work in the solar and electronics industries. Since then we have been refurbishing and upgrading the facility to produce commercial quantities of Carbon-14. In June 2023, we entered into a tolling agreement with a North American customer for the entire capacity of this facility, under which we will supply the customer with C-14 enriched to 85%.
| 7 |
| Table of Contents |
Demonstrate the capability to produce C-14, Mo-100, and Si-28 using the ASP technology and capitalize on the opportunity to solve many supply chain challenges that currently exist.
We intend to demonstrate the capability to produce C-14, Mo-100, and Si-28 at a scale that can support anticipated customer demand for all three isotopes.
Historically, Russia has been the sole supplier of C-14, which is used as a tracer in the development of new pharmaceuticals and agrochemicals. The supply chain has been inherently fragile with inconsistent service. Subject to the supply of feedstock from our customer, we intend to start the enrichment of C-14 during 2024.
Mo-100 as an alternative and potentially more convenient production route for Tc-99m used in nuclear medical diagnostic procedures. Mo-99’s decay product, technetium-99m (Tc-99m), is used in 80-85% of the world’s single-photon emission computed tomography (SPECT) procedures, which is used to diagnose heart disease and cancer, to study organ structure and function, and to perform other critical medical applications. We intend to offer our Mo-100 to customers who may convert Mo-100 into Mo-99 or directly into Tc-99m, and we believe that the use of Mo-100 in this way will be an attractive alternative route to the production of Tc-99m for several reasons.
| · | Only a small number of major reactors located around the world (e.g., Australia, Belgium, the Netherlands, and South Africa) produce large-scale amounts of Mo-99. These reactors are taken off-line periodically for refueling and maintenance and go off-line on an unscheduled basis due to the need for extended repairs, which results in a global Mo-99 supply chain that is lengthy, complex, and prone to interruption and has experienced supply shortages. Customers that could use and stockpile Mo-100 due to its stable profile when compared to Mo-99 would not have to manage the periodic shortages and supply chain challenges related to Mo-99. |
|
|
|
| · | Mo-99 (a radioisotope with a 66-hour half-life) decays and loses activity in transit, so it must be moved through the supply chain quickly to minimize decay losses, and it cannot be stockpiled. Mo-100 (a stable isotope of molybdenum) will not decay in transit, so the supply chain would not depend on elapsed time from the production of Mo-100 to the delivery of a Tc-99m dose to a hospital or clinic. |
|
|
|
| · | Mo-99 (with decay product Tc-99m) must be shipped in shielded transport containers that comply with the regulatory requirements for the safe transport of radioactive material. Mo-100 is stable (non-radioactive) and, therefore, does not have the same handling and shipping requirements. |
Isotopically enriched silicon is regarded as a promising material for semiconductor quantum information due to its very long coherence times and its compatibility with the readily available industrial platform. We believe that the ASP technology is ideally suited to the production of this isotope because it has the ability to enrich molecules of low molecular mass. Other electronic gasses that can likely be enriched using ASP Technology include disilane and germane.
Continue identifying potential offtake customers and strategic partners for our isotopes.
We have already seen significant interest from potential offtake customers for the isotopes that we intend to produce. In November 2022, we entered a 25-year supply agreement for highly enriched Mo-100 with BRICEM (Beijing Research Institute of Chemical Engineering Metallurgy). The contract has a value of up to $27.0 million per annum. In July 2023, we entered into a supply agreement for a highly enriched metal with a U.S. customer. This contract has an annual sales value of $9 million. We have had or are currently in active dialogue with many other potential customers who could use the entire anticipated annual capacity of an initial plant. In June 2023, we entered into a tolling agreement with a Canadian customer for the entire capacity of our C-14 production facility. We are currently in discussions with potential customers that have an interest in entering into long-term supply agreements for kilogram quantities of Si-28 and larger quantities of Xe-129, Ge 72, Ge-74, Zn-68, and Cl-37.
Demonstrate the capability to produce high-assay low-enriched uranium (HALEU) using Quantum Enrichment and meet anticipated demand for the new generation of HALEU-fueled small modular reactors and advanced reactor designs that are now under development for commercial and government uses.
We plan to begin research and development for the enrichment of uranium to demonstrate our capability to produce HALEU using Quantum Enrichment technology. We anticipate a future demand for HALEU for the new generation of HALEU-fueled small modular reactors (SMRs) and advanced reactor designs that are now under development for commercial and government uses. SMRs are viewed as being cheaper, safer, and more versatile than traditional large-scale nuclear reactors, and development of the new technology is receiving considerable funding from the U.S. Department of Energy, as well as from the governments of other countries. There is currently no commercial production of HALEU in the United States. We are currently conducting a feasibility study with respect to constructing an enrichment facility in either South Africa or the United Kingdom. We are currently in discussions with both the UK Atomic Energy Authority, UK Office of Nuclear Regulation (ONR), Nuclear Energy Corporation of South Africa (NECSA) as well as the South African Department of Mineral Resources and Energy (DMRE) pursuing an approval to conduct nuclear research in either of these countries. We would need to obtain approval from one of these regulatory bodies in order to proceed with our nuclear research. Alongside our talks with regulators, we are currently discussing with multiple counterparties involved in the SMR space to produce HALEU to further their research efforts and, later, their commercial endeavors.
| 8 |
| Table of Contents |
Initiate the R&D efforts of the Quantum Enrichment Process via the Company’s subsidiary, Enlightened Isotopes, for the enrichment of Ytterbium 176
We anticipate furthering our commercial development of the Quantum Enrichment process through the development of enriching Ytterbium-176 for the medical oncology market. Enriched Ytterbium-176 can be irradiated to produce Lutetium-177, which has been identified for use in oncology, particularly in targeted radionuclide therapy (TRT). TRT is used in the treatment of various types of cancers, including neuroendocrine tumors, prostate cancer, and bone metastases, among others. There are 11 ongoing clinical trials studying Lutetium-177 PSMA-617 in patients with metastatic castration-resistant prostate cancer. We have obtained all necessary licenses within South Africa to proceed with the commercial development of this product. We anticipate that this continued R&D and later clinical applicability will help to refine the Quantum Enrichment process for broader commercial applicability in the future.
Demonstrate the effectiveness and value in the use of Mo-100 and other stable isotopes in the downstream radiopharmacy market, after acquiring 51% ownership interest in PET Labs, the leading radiopharmacy in South Africa. This investment will address the radioisotope needs of South Africa as well as certain neighboring countries.
Under the terms of a Share Purchase Agreement, dated October 30, 2023, we acquired 51% of the issued share capital of Pet Labs Pharmaceuticals Proprietary Limited, a company incorporated in the Republic of South Africa (“PET Labs”). PET Labs is a South African radiopharmaceutical operations company, dedicated to nuclear medicine and the science of radiopharmaceutical production. As a result of this transaction, we entered into the downstream radiopharmacy market that we intend to service in the future. This transaction will help provide the market with adequate proof of concept of the value of utilizing Mo-100 in downstream SPECT imaging procedures while providing supply chain stability to the region of South Africa and neighboring countries. We intend to expand PET Labs’ existing operations by adding two new cyclotrons to its service footprint, enabling the company to properly expand its other revenue generation mediums, which is anticipated to drive free cash flow to the company.
Our Strengths
ASP technology initially developed by Klydon and further developed by ASPI (S.Africa).
The aerodynamic separation technique has its origins in the South African uranium enrichment program in the 1980s, and the ASP technology has been developed during the last 18 years by the scientists at Klydon. To date, the scientists at Klydon have constructed two ASP plants for the enrichment of oxygen-18 and silicon-28 in Pretoria, South Africa, which were commissioned in October 2015 and July 2018, respectively. While the technology has not yet been used to enrich either Molybdenum or Uranium or heavier isotopes, we believe the success of the enrichment process for oxygen-18 and silicon-28 has demonstrated the efficacy and commercial scalability of the ASP technology. If our research and development is successful (and subject to obtaining applicable regulatory approvals and appropriate licenses), we plan to commercialize many different isotopes produced using the ASP technology. To date, we have completed the construction of one isotope enrichment facility, but we have not yet produced any commercial quantities of isotopes and we have not yet demonstrated the ability to produce any isotope in commercial quantities using ASP technology.
Extensive R&D Experience in Aerodynamic Separation Technology and Processes.
Subject to successful research and development, our ASP technology has the potential to produce many different types of isotopes. Klydon has spent the last 18 years and tens of millions of dollars developing the aerodynamic separation technique used in the ASP technology, generating critical trade secrets. We believe our competitors lag behind us in terms of the technical expertise of our senior management and the know-how contained in the aerodynamic separation technique and will be unable to replicate the expected results of the ASP technology, even as we expect to continue to improve the existing technology and processes. Additionally, the high capital costs of development of proprietary technologies, significant lead times required to construct new enrichment facilities, as well as stringent regulatory and operating requirements applicable to enrichment facilities, adds to the significant barriers to entry for smaller competing market participants.
| 9 |
| Table of Contents |
ASP technology is a flexible platform with the potential to produce many different isotopes that could serve a large addressable markets.
ASP technology is a flexible platform, compact in size and weight, and could be easily scaled to an industrial level with number of separation devices added in parallel. The ASP technology also has few moving parts, with low capital and operating costs in comparison to alternatives. The technology is particularly efficient at enriching isotopes of low atomic mass. We believe that, assuming receipt of required regulatory approvals and governmental permits, the ASP technology can be deployed quickly and with a relatively minimal capital cost, to enrich many different isotopes that we believe consumers require both today and in the future in end markets such as healthcare, technology and energy.
ASP technology is designed to be low cost, low energy, and environmentally friendly.
We recently completed the construction of our first isotope enrichment facility using ASP technology located in Pretoria, South Africa. The ASP technology is designed to be scalable, low cost, low energy, and environmentally friendly, with no radioactive waste or hazardous materials produced in the process and planned arrangements to reuse chemical by-products.
Experienced team
Our board of directors and advisers have specialized expertise in isotope enrichment, R&D, technology, plant development, and manufacturing. Dr. Einar Ronander, who serves as Chief Scientific Adviser to our board of directors, and Dr Hendrik Strydom, one of our directors, previously co-founded Klydon. The scientific team at Klydon combined has decades of experience in research and development of isotope enrichment and amassed deep knowledge in the field.
Our board of directors and our management team also have broad experience and successful track records in fusion technology and fusion materials, biopharmaceutical research, chemicals, manufacturing and commercialization, as well as in business, operations, and finance. Our board of directors’ and management team’s experience was gained at leading companies and financial institutions that include, Bear Stearns, Deutsche Bank, Highbridge Capital, Investec Bank, Morgan Stanley and Soros Fund Management.
Technical Background
What are Isotopes?
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers (mass numbers) due to different numbers of neutrons in their nuclei. While all isotopes of a given element have almost the same chemical properties, they have different atomic masses and physical properties.
The number of protons within the atom’s nucleus is called atomic number and is equal to the number of electrons in the neutral (non-ionized) atom. Each atomic number identifies a specific element, but not the isotope; an atom of a given element may have a wide range in its number of neutrons. The number of nucleons (both protons and neutrons) in the nucleus is the atom’s mass number, and each isotope of a given element has a different mass number. For example, carbon-12, carbon-13, and carbon-14 are three isotopes of the element carbon with mass numbers 12, 13, and 14, respectively. The atomic number of carbon is 6, which means that every carbon atom has 6 protons so that the neutron numbers of these isotopes are 6, 7, and 8 respectively.
There are 23 isotopes of Silicon, all of which have 14 protons and 14 neutrons but have between 8 and 30 neutrons. The table below shows a selection of those isotopes. Three isotopes are stable which have mass numbers of 28, 29 and 30 which have 14, 15 and 16 neutrons respectively. The other 20 isotopes are radioactive and decay with short half-lives and are therefore do not typically exist in naturally occurring silicon. In naturally occurring silicon, the isotope with atomic mass of 28 is usually the most abundant, typically accounting for approximately 92.22% of the material. The isotope with atomic mass of 29 typically accounts for 4.69% of the material and the isotope with atomic mass of 30 typically accounts for 3.09% of the material.
Molybdenum has 33 known isotopes, ranging in atomic mass from 83 to 115, as well as four metastable nuclear isomers. Seven isotopes occur naturally, with atomic masses of 92, 94, 95, 96, 97, 98, and 100. All unstable isotopes of molybdenum decay into isotopes of zirconium, niobium, technetium, and ruthenium.
| 10 |
| Table of Contents |
Uranium is a naturally occurring radioactive element that has no stable isotope. It has two primordial isotopes, uranium-238 and uranium-235, which have long half-lives and are found in appreciable quantity in the Earth’s crust. The decay product, uranium-234 is also found. Other isotopes such as uranium-233 have been produced in breeder reactors. In addition to isotopes found in nature or nuclear reactors, many isotopes with far shorter half-lives have been produced, ranging from U-214 to U-242 (with the exception of U-220 and U-241). The standard atomic weight of natural uranium is 238.02891 with 99.27% of naturally occurring uranium being the isotope with an atomic mass of 238.
Selected isotopes of Silicon |
| Selected isotopes of Molybdenum |
| Selected isotopes of Uranium | ||||||||||||||||||||||||||||||
Nuclide |
| Protons |
| Neutrons |
| Isotopic Mass |
| Half Life |
| Natural abundance |
| Nuclide |
| Protons |
| Neutrons |
| Isotopic Mass |
| Half Life |
| Natural abundance |
| Nuclide |
| Protons |
| Neutrons |
| Isotopic Mass |
| Half Life |
| Natural abundance |
22 |
| 14 |
| 8 |
| 22.036 |
| 29 ms |
|
|
| 91 |
| 42 |
| 49 |
| 90.912 |
| 15.49 min |
|
|
| 225 |
| 92 |
| 133 |
| 225.029 |
| 62 ms |
|
|
23 |
| 14 |
| 9 |
| 23.025 |
| 42.3 ms |
|
|
| 92 |
| 42 |
| 50 |
| 91.907 |
| Stable |
| 14.65% |
| 226 |
| 92 |
| 134 |
| 226.029 |
| 269 ms |
|
|
24 |
| 14 |
| 10 |
| 24.012 |
| 140 ms |
|
|
| 93 |
| 42 |
| 51 |
| 92.907 |
| 4000 y |
|
|
| 227 |
| 92 |
| 135 |
| 227.031 |
| 1.1 m |
|
|
25 |
| 14 |
| 11 |
| 25.004 |
| 220 ms |
|
|
| 94 |
| 42 |
| 52 |
| 93.905 |
| Stable |
| 9.19% |
| 228 |
| 92 |
| 136 |
| 228.031 |
| 9.1 m |
|
|
26 |
| 14 |
| 12 |
| 25.992 |
| 2.245 s |
|
|
| 95 |
| 42 |
| 53 |
| 94.906 |
| Stable |
| 15.87% |
| 229 |
| 92 |
| 137 |
| 229.034 |
| 57.8 m |
|
|
27 |
| 14 |
| 13 |
| 26.987 |
| 4.15 s |
|
|
| 96 |
| 42 |
| 54 |
| 95.905 |
| Stable |
| 16.67% |
| 230 |
| 92 |
| 138 |
| 230.034 |
| 20.23 d |
|
|
28 |
| 14 |
| 14 |
| 27.977 |
| Stable |
| 92.22% |
| 97 |
| 42 |
| 55 |
| 96.906 |
| Stable |
| 9.58% |
| 231 |
| 92 |
| 139 |
| 231.036 |
| 4.2 d |
|
|
29 |
| 14 |
| 15 |
| 28.977 |
| Stable |
| 4.69% |
| 98 |
| 42 |
| 56 |
| 97.905 |
| Stable |
| 24.29% |
| 232 |
| 92 |
| 140 |
| 232.037 |
| 68.9 y |
|
|
30 |
| 14 |
| 16 |
| 29.974 |
| Stable |
| 3.09% |
| 99 |
| 42 |
| 57 |
| 98.908 |
| 2.75 d |
|
|
| 233 |
| 92 |
| 141 |
| 233.04 |
| 1.592 e5 y |
| Trace |
31 |
| 14 |
| 17 |
| 30.975 |
| 157.36 min |
|
|
| 100 |
| 42 |
| 58 |
| 99.907 |
| Stable |
| 9.74% |
| 234 |
| 92 |
| 142 |
| 234.041 |
| 2.455 e5 y |
| Trace |
32 |
| 14 |
| 18 |
| 31.974 |
| 153 y |
| trace |
| 101 |
| 42 |
| 59 |
| 100.910 |
| 14.61 m |
|
|
| 235 |
| 92 |
| 143 |
| 235.044 |
| 7.038 e8 y |
| 0.72% |
33 |
| 14 |
| 19 |
| 32.978 |
| 6.18 s |
|
|
| 102 |
| 42 |
| 60 |
| 101.910 |
| 11.3 m |
|
|
| 236 |
| 92 |
| 144 |
| 236.046 |
| 2.342 e7 y |
| Trace |
34 |
| 14 |
| 20 |
| 33.979 |
| 2.77 s |
|
|
| 103 |
| 42 |
| 61 |
| 102.913 |
| 67.5 s |
|
|
| 237 |
| 92 |
| 145 |
| 237.049 |
| 6.752 d |
| Trace |
35 |
| 14 |
| 21 |
| 34.985 |
| 780 ms |
|
|
| 104 |
| 42 |
| 62 |
| 103.914 |
| 60 s |
|
|
| 238 |
| 92 |
| 146 |
| 238.051 |
| 4.468 e9 y |
| 99.27% |
36 |
| 14 |
| 22 |
| 35.987 |
| 450 ms |
|
|
| 105 |
| 42 |
| 63 |
| 104.917 |
| 35.6 s |
|
|
| 239 |
| 92 |
| 147 |
| 239.054 |
| 23.45 m |
|
|
37 |
| 14 |
| 23 |
| 36.993 |
| 90 ms |
|
|
| 106 |
| 42 |
| 64 |
| 105.918 |
| 8.73 s |
|
|
| 240 |
| 92 |
| 148 |
| 240.057 |
| 14.1 h |
| Trace |
38 |
| 14 |
| 24 |
| 37.996 |
| 90 ms |
|
|
| 107 |
| 42 |
| 65 |
| 106.922 |
| 3.5 s |
|
|
| 242 |
| 92 |
| 150 |
| 242.063 |
| 16.8 m |
|
|
Methods of Separation and Enrichment of Isotopes
Isotope enrichment is the process of concentrating specific isotopes of a chemical element by removing other isotopes. During the last century, a number of different methods have been developed to separate and enrich isotopes. The current separation or enrichment processes are based either on the atomic weight of the isotope, small differences in chemical reaction rates produced by different atomic weights or are based on properties not directly connected to atomic weight such as nuclear resonances.
| 11 |
| Table of Contents |
Diffusion
Often performed on gases, but also on liquids, the diffusion method relies on the fact that in thermal equilibrium, two isotopes with the same energy will have different average velocities. The lighter atoms (or the molecules containing them) will travel more quickly and be more likely to diffuse through a membrane. The difference in speeds is proportional to the square root of the mass ratio, so the amount of separation is small, and many cascaded stages are needed to obtain high purity. This method is expensive due to the work needed to push gas through a membrane and the many stages necessary.
Centrifugal
Centrifugal methods rapidly rotate the material allowing the heavier isotopes to go closer to an outer radial wall. This too is often done in gaseous form using a Zippe-type centrifuge.
A Zippe-type centrifuge relies on the force resulting from centripetal acceleration to separate molecules according to their mass, and can be applied to most fluids. The dense (heavier) molecules move towards the wall and the lighter ones remain close to the center. The centrifuge consists of a rigid body rotor rotating at full period at high speed. Concentric gas tubes located on the axis of the rotor are used to introduce feed gas into the rotor and extract the heavier and lighter separated streams. For U-235 production, the heavier stream is the waste stream and the lighter stream is the product stream. Modern Zippe-type centrifuges are tall cylinders spinning on a vertical axis, with a vertical temperature gradient applied to create a convective circulation rising in the center and descending at the periphery of the centrifuge. Diffusion between these opposing flows increases the separation by the principle of countercurrent multiplication.
In practice, since there are limits to how tall a single centrifuge can be made, several such centrifuges are connected in series. Each centrifuge receives one input and produces two output lines, corresponding to light and heavy fractions. The input of each centrifuge is the output (light) of the previous centrifuge and the output (heavy) of the following stage. This produces an almost pure light fraction from the output (light) of the last centrifuge and an almost pure heavy fraction from the output (heavy) of the first centrifuge.
Electromagnetic
Electromagnetic separation is mass spectrometry on a large scale, so it is sometimes referred to as mass spectrometry. It uses the fact that charged particles are deflected in a magnetic field and the amount of deflection depends upon the particle’s mass. It is very expensive for the quantity produced, as it has an extremely low throughput, but it can allow very high purities to be achieved. This method is often used for processing small amounts of pure isotopes for research or specific use (such as isotopic tracers), but is impractical for industrial use.
Laser
In this method, a laser is tuned to a wavelength which excites only one isotope of the material and ionizes those atoms preferentially. The resonant absorption of light for an isotope is dependent upon its mass and certain hyperfine interactions between electrons and the nucleus, allowing finely tuned lasers to interact with only one isotope. After the atom is ionized it can be removed from the sample by applying an electric field. This method is often abbreviated as AVLIS (atomic vapor laser isotope separation). This method has only recently been developed as laser technology has improved, and is currently not used extensively.
Chemical Methods
Although isotopes of a single element are normally described as having the same chemical properties, this is not strictly true. In particular, reaction rates are very slightly affected by atomic mass. Techniques using this are most effective for light atoms such as hydrogen. Lighter isotopes tend to react or evaporate more quickly than heavy isotopes, allowing them to be separated. This is how heavy water is produced commercially.
Gravity
Isotopes of carbon, oxygen, and nitrogen can be purified by chilling these gases or compounds nearly to their liquefaction temperature in very tall (200 to 700 feet (61 to 213 m)) columns. The heavier isotopes sink and the lighter isotopes rise, where they are easily collected.
| 12 |
| Table of Contents |
The Aerodynamic Separation Process (ASP) Technology
ASP technology is proprietary technology licensed from Klydon which succeeds earlier work, first detailed in the scientific media in the mid-1970s, relating to an industrial scale enrichment plant for uranium that was constructed utilizing the so-called “stationary-wall centrifuge”. The original technology was highly energy consuming and was not able to compete on an economic basis with other methods of isotope separation. The innovative development of the ASP technology over the past 18 years has culminated in a more advanced separation device that we believe can compete on a commercial scale with other methods of isotope separation. The ASP separation device separates both gas species and isotopes in a volatile state via an approximate flow pattern as shown below.
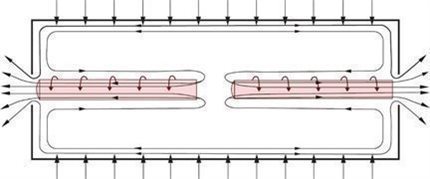
Gas flow pattern inside ASP separation device.
The ASP enrichment process uses an aerodynamic technique similar to a stationary wall centrifuge. The isotope material in raw gas form enters the stationary tube at high speed by tangential injection through finely placed and sized openings in the surface of the tube. The gas then follows a flow pattern that results in two gas vortexes occurring around the geometrical axis of the separator. The isotope material becomes separated in the radial dimension as a result of the spin speed of the isotope material reaching several hundred meters per second. An axial mass flow component in each tube feeds isotope material to the respective ends of the separator where the collection of the portions of isotope material is accomplished.
The advantages of ASP technology are as follows:
| · | No moving parts, with low capital and operating costs in comparison to alternatives. |
|
|
|
| · | Compact in size and weight. |
|
|
|
| · | Easily scaled to industrial level with number of separation devices added in parallel. |
|
|
|
| · | The separation process occurs inside a closed cylindrical container and is a volume technology, i.e., the process efficiency is not affected by poisoning of surface contaminates as is the case for surface separation processes. |
|
|
|
| · | ASP operates very efficiently at molecular masses below 100 atomic mass units, unlike other separation processes which are more efficient higher masses, which ASP can achieve equally well or to a superior degree. |
|
|
|
| · | ASP easily separates hydrogen gas from other gas components, e.g., harvesting hydrogen gas from carbon monoxide and carbon dioxide and altering the ratio of syngas mixture. |
|
|
|
| · | With the right material choice ASP handles even the most corrosive gases. |
|
|
|
| · | ASP can separate any isotopes that have a gaseous or volatile chemical compound. |
|
|
|
| · | Most of the subsystems are procured from off-the-shelf components. |
|
|
|
| · | An ASP plant can be constructed in any country that adheres to the International Atomic Energy Agency (IAEA) protocols for the protection of dual use technology. |
| 13 |
| Table of Contents |
ASP Plant Configuration
The figure below shows a schematic of an ASP cascade in operation. The cascade consists of several enrichment stages, connected in a 1-up-1-down cascade configuration. The stages can be grouped into segments. (This method of organizing stages is not reflected in the figure)
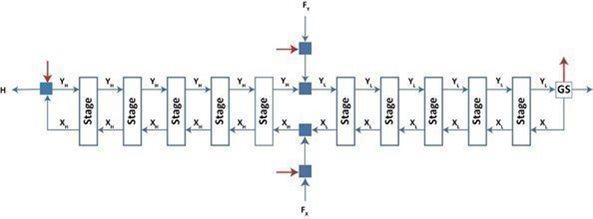
The bold blue arrows represent flows of the element into and out of the cascade:
| · | H is the product, enriched in the isotope |
|
|
|
| · | L is the tails, stripped of the isotope |
|
|
|
| · | F = FX + FY is the feed stream at natural isotopic composition: |
|
|
|
| · | FX is the feed into the product stream of an adjoining stage. |
|
|
|
| · | FY is the feed into the tails stream of an adjoin |
Each stage in the cascade is operated in one of two configurations:
| (1) | A net backward flow of the isotope: Xi > Yi. These stages are referred to as “product”, situated in the so-called “product cascade section”, and their flows are marked with an “H” subscript. |
|
|
|
| (2) | A net forward flow of isotope: Xi < Yi. These stages are referred to as “tails”, situated in the so-called “tails cascade section”, and their flows are marked with an “L” subscript. |
The red arrows represent the addition or extraction of carrier gas from the process. The arrows have been added for clarity and orientation, but the mass flows of the carrier gas will be ignored in the rest of the discussion as it pertains to the isotope mass flows only (as represented by the blue arrows). The carrier gas mass flows can be superimposed on any isotope mass balance using the molar mass characteristics of the ASP stages (see below).
The block marked “GS” represents the gas separator: a piece of equipment used to separate the carrier gas from the element of interest to the degree necessary to provide a suitable reflux stream to the tails cascade section.
The blue squares are simply suitable areas where streams can be split or mixed.
An ASP stage is characterized by functions of Y, the flow of isotope in its tails stream. The characteristics of interest are:
| · | α(Y): the separation factor between the tails and product streams. |
|
|
|
| · | MY(Y): the molar mass of the tails stream. |
|
|
|
| · | MX(Y): the molar mass of the product stream. |
|
|
|
| · | P(Y): the stage’s power usage. |
|
|
|
| · | X(θ,Y): the flow of Zinc in the product stream, where θ = Y/(X+Y) is the cut defined in terms of isotope flows. |
Note the following:
| · | α is the ratio of the tails and product stream abundance ratios. |
|
|
|
| · | Y, X(θ,Y) and α(Y) describe the stage’s behaviour with regards to Zinc, while MY(Y) and MX(Y) defines its behaviour with regards to the carrier gas. |
|
|
|
| · | P, the stage’s power usage, depends on the ASP separator, but also on factors such as compressor efficiency, friction losses etc. It is therefore a partial function of stage design. |
|
|
|
| · | It is possible to define Pmin, the theoretical minimum energy usage of a stage, by assuming 100% efficient compressors and no losses in the stage. Pmin is a function of the ASP separator only. In practice P is a more useful metric, as the contribution of compressor inefficiencies to power consumption is significant. |
|
|
|
| · | Except for X, the stage’s characteristics are not defined in terms of the cut θ, as they are simply not sensitive to it above a certain lower limit θmin. In practice θmin is small enough that it has no influence on the normal operating envelope of the stage. |
|
|
|
| · | X is per definition a function of Y via θ as indicated. |
| 14 |
| Table of Contents |
The cut of an ASP stage can be dynamically adjusted to any value larger than θmin, allowing its operating point to be changed online during production.
All stages in the product cascade section are operated at the same point < XH,YH >, where XH > YH, ensuring that a net backward flow of the process element, H = XH — YH is achieved. This corresponds to a cut of less than 50% and ensures a positive flow of enriched product.
All stages in the tails cascade section are operated at the same point < XL,YL >, where XL < YL, ensuring that a net backward flow of the process element, L = XL — YL is achieved. This corresponds to a cut of more than 50% and ensures a positive flow of stripped tails.
Depending on the production requirements of the cascade the product and tails section operation points can be moved relative to each other during production, obtaining different combinations of H and L (and therefore different feeds F = H + L). The smaller H (or L) is chosen, the closer the product (or tails) section cut moves to 50%. If all stages are operated at a cut of 50%, the cascade is operated at full reflux, no product, tails, or feed streams are present, and the maximum process element concentration gradient will exist.
ASP Technology In Use
To date, the scientists at Klydon have constructed two ASP plants for the enrichment of oxygen-18 and silicon-28 in Pretoria, South Africa, which were commissioned in October 2015 and July 2018, respectively,. We believe the success of the enrichment of oxygen-18 and silicon-28 has demonstrated the efficacy and commercial scalability of the ASP technology. We are currently constructing two enrichment plants, which, if successful, will be able to produce a range of isotopes, including but not limited to C-14, Mo-100 and Si-28.
Nuclear Medicine
Nuclear medicine is a medical specialty that utilizes radioactive isotopes, referred to as radionuclides, to diagnose and treat disease. These radionuclides are incorporated into radiopharmaceuticals and introduced into the body by injection, swallowing, or inhalation. Physiologic/metabolic processes in the body concentrate the tracers in specific tissues and organs; the radioactive emissions from the tracers can be used to noninvasively image these processes or kill cells in regions where radionuclides have concentrated.
Other types of noninvasive diagnostic procedures — for example, computed tomography (CT) and magnetic resonance imaging (MRI) — can detect anatomical changes in tissues and organs as the result of disease. Nuclear medicine procedures can often detect the physiological and metabolic changes associated with disease before any anatomical changes occur. Such procedures can be used to identify disease at early stages and evaluate patients’ early responses to therapeutic interventions.
Single Photon Emission Computed Tomography (SPECT) generates three-dimensional (3D) images of tissues and organs using radionuclides that emit gamma rays; the most used radionuclide is Technitium-99m (Tc-99m), often referred to as the ‘work-horse’ of nuclear medicine. Individual gamma rays emitted from the decay of these radionuclides (i.e., single photon emissions) are detected using a gamma camera. This camera technology is used to obtain two-dimensional (2D) images; 3D SPECT images are computer generated from many 2D images recorded at different angles.
Positron Emission Tomography (PET) generates 3D images of tissues and organs using tracers that emit positrons (i.e., positive electrons): for example, fluorine-18 (F-18). Annihilation reactions between the positrons from these radionuclides and electrons present in tissues and organs produce photons. (Two photons are emitted simultaneously for each annihilation reaction and essentially travel in opposite directions.) The photon pairs are detected with a camera having a ring of very fast detectors and electronics. PET images generally have a higher contrast and spatial resolution than do SPECT images. However, PET equipment is more expensive and therefore not as widely available as SPECT equipment. Additionally, most PET tracers have short half-lives (e.g., nitrogen-13 (N-13): 10 minutes, carbon-11 (C-11): 20 minutes, and F-18: 110 minutes), so they must be produced close to their point of use.
| 15 |
| Table of Contents |
Technetium-99m (Tc-99m)–the most widely used radioisotope in Nuclear Imaging
Tc-99m is used in approximately 80 percent of all nuclear medicine procedures performed worldwide each year.
Tc-99m is a particularly useful imaging radionuclide because it:
| · | Has a sufficiently long half-life (~6 hours) to be usable in nuclear medicine procedures. |
|
|
|
| · | Emits energetic gamma rays (140 kiloelectron volts [keV]) that can be detected efficiently with widely available camera technologies. |
|
|
|
| · | Provides low patient doses for some procedures because of its short half-life and lack of alpha or beta radiations. |
Tc-99m-based radiopharmaceuticals are used to diagnose disease in many tissue and organ systems, including bone, brain, heart, kidneys, liver, and lungs. About 50 percent of Tc-99m utilization in the United States is in nuclear cardiology, predominantly for myocardial perfusion imaging which images blood flow through heart muscle.
Because Tc-99m has a half life of just 6 hours, it cannot be stored or shipped long distances and it is currently produced using a technetium generator, which contains Molybdenum-99 which has a half-life of about 66-hours. In the reactor, Mo-99 decays to Tc-99m by emitting a beta particle (an electron). About 88 percent of the decays produce Tc-99m, which subsequently decays to the ground state, Tc-99g, by emitting a gamma ray. About 12 percent of the decays produce Tc-99g directly. Tc-99g decays to stable (i.e., nonradioactive) ruthenium-99 (Ru-99) after emitting a beta particle.
Technetium generators are systems that store Mo-99 and allow its decay product, Tc-99m, to be recovered for use. Most technetium generators are designed to be used with high-specific-activity Mo-99 (>1,000 Ci/g) produced by U-235 fission. The generator consists of an alumina (Al2O3) column having the diameter of a large pencil along with associated filters and tubing for obtaining Tc-99m.
This apparatus is installed into radiation-shielded packages for shipment to Tc-99m suppliers. The generator includes both the package and its contained apparatus. Technetium generators can contain from 1 to 19 Ci of Mo-99, matched to address the needs and workloads of Tc-99m suppliers.
It takes 18-24 hours to prepare technetium generators for shipment. Preparation involves loading the molybdate solution onto the columns and sterilizing them; installing the columns, tubing, and filters into the shielded generator package; and packaging the generators for shipment. Tc-99m generators are typically shipped to Tc-99m suppliers within a day of their manufacture. Generators are shipped in regulatory-compliant boxes. The delivery methods can be air, ground, or a combination of both depending on customer location and contracted transportation network.
The Mo-99 Market
The global medical community depends on a reliable supply of the radioisotope Mo-99 for nuclear medical diagnostic procedures. As previously described, Mo-99’s decay product, technetium-99m (Tc-99m), is used in over 40,000 medical procedures in the United States each day to diagnose heart disease and cancer, to study organ structure and function, and to perform other important medical applications.
In 2020, it is estimated (by Future Market Insights Inc, a global market research firm), that the Molybdenum 99 market generated revenues of approximately $3.8 billion. North America accounted for almost half of the Mo-99 demand. Approximately 62% of Mo-99 was used in hospitals while approximately 38% of Mo-99 use was in diagnostic centers.
The Mo-99 Supply Chain
The global Mo-99 supply chain is inherently fragile. The fragility stems primarily from two factors:
| 1. | Mo-99 and its daughter isotope Tc-99m have short half-lives (66 and 6 hours, respectively) and therefore cannot be stockpiled. These radioisotopes need to be produced and delivered to the supply chain on a weekly or more frequent basis. |
|
|
|
| 2. | Global supply of Mo-99 currently relies on a small number of aging reactors worldwide and a small number of suppliers. |
The current Mo-99 supply chain is also lengthy and prone to interruption throughout its course.
Recent Government Efforts to Increase Mo-99 Availability
Given the regular supply side shortages in the Mo-99 market, and widely anticipated shutdown of many of the current reactors, there is considerable focus on alternative methods of Tc-99m production. In 2012, Congress passed the American Medical Isotopes Production Act (AMIPA), which directed the National Nuclear Security Administration (NNSA) to establish a technology-neutral program to support the establishment of domestic supplies of Mo-99 without the use of HEU. NNSA has implemented this by competitively awarding 50%/50% cost-shared cooperative agreements to commercial entities and providing funds to the Department of Energy’s (DOE) National Laboratories to support development of non-HEU Mo-99 production technologies.
| 16 |
| Table of Contents |
NNSA currently manages cooperative agreements with three U.S. companies, all developing diverse Mo-99 production technologies:
| · | NorthStar Medical Radioisotopes, LLC (Beloit, Wisconsin) |
| o | Neutron capture technology using molybdenum-98 targets |
| o | Accelerator-based technology using molybdenum-100 targets |
| · | SHINE Technologies, LLC (Janesville, Wisconsin) |
| o | Accelerator with fission technology to produce Mo-99 with an LEU solution target |
| · | Niowave, Inc. (Lansing, Michigan) |
| o | Superconducting electron linear accelerator with fission technology to produce Mo-99 with LEU targets |
Mo-100 as an Alternative Intermediate to Produce Mo-99 and Tc-99m
Mo-100 is a stable isotope of molybdenum. Naturally occurring molybdenum contains approximately 9.74% molybdenum-100. When highly enriched so that the Molybdenum contains >95% of the Mo-100 isotope, it can be used to produce either Mo-99 or Tc-99 via either photon-induced transmutation of Mo-100 into Mo-99 or via proton bombardment of Mo-100 into Tc-99m. The use of particle accelerators for the production of Mo-99 and direct production of Tc-99m has been studied extensively and the use of a particle accelerator conveys certain advantages and disadvantages. Accelerators produce ion beams and accelerate ions to higher energies by using oscillating electromagnetic fields. The accelerated particle beams have the capability of irradiating specific targets to produce Mo-99 and/or Tc-99m.
We intend to offer our Mo-100 to customers that may convert Mo-100 into Mo-99 or Mo-100 directly into Tc-99m. We believe that customers will be able to convert Mo-100 into Mo-99 using a cyclotron or a linear accelerator. The Mo-99 can then be converted into Tc-99m using a technetium generator. The technetium generators that are currently available will likely require some modifications in order to use the Mo-99 that has been produced via a cyclotron or a linear accelerator. These modifications will likely mean that new generator will require approval by healthcare regulators such as the Food and Drug Administration (FDA) in the United States and the European Medicines Agency (EMA) in Europe.
Customers may convert Mo-100 directly into Tc-99m using a cyclotron, which would eliminate the need for a technetium generator. To date, only one healthcare regulator (Health Canada) has approved the use of Tc-99m that has been directly produced from Mo-100 in a low powered cyclotron. We believe it is likely that healthcare regulators in other countries will also require clinical data to support the use of Tc-99m that is produced directly from Mo-100.
ASP Technology for Carbon-14 Enrichment
C-14 is a radioactive isotope of carbon with a half-life of 5,700 years that has a natural abundance of 1 part per trillion. The different isotopes of carbon do not differ appreciably in their chemical properties. This resemblance is used in chemical and biological research, in a technique called carbon labelling: carbon-14 atoms can be used to replace nonradioactive carbon, in order to trace chemical and biochemical reactions involving carbon atoms from any given organic compound.
Carbon-14 could be obtained from waste by-products in certain nuclear reactors. In June 2023, we entered into a multi-year supply agreement with a Canadian Customer for the supply of Carbon-14, which will be produced from our facility that was completed in March 2023. The customer will supply carbon-14 in the form of carbon-dioxide gas. We will then convert the carbon dioxide gas into methane under a chemical converting contract entered in June 2023. We will then enrich the methane to greater than 85% C-14 under a tolling agreement, also entered in June 2023. Finally, we will convert the enriched methane back into enriched carbon dioxide under a chemical converting contract. The tolling agreement has a minimum “take or pay” amount of approximately $2.5 million per year, supported by a bank letter of guarantee. In September 2023, we entered into a Memorandum of Understanding (MOU) with the same customer to separate Deuterium and Tritium currently stored at nuclear sites within Canada. The timing and commercial implications of this MOU are subject to future agreement between the parties.
| 17 |
| Table of Contents |
ASP Technology for Silicon-28 Enrichment
Si-28 is a stable isotope of silicon. Isotopically enriched Si-28 is regarded as an ideal host material for semiconducting quantum computing due to the lack of Si-29 nuclear spins. The presence of Si-29 in concentrations above 500 parts per million (ppm) (0.05%) prevents effective performance. The lower the concentration of Si-29, the better a silicon quantum processor will perform in terms of computational power, accuracy and reliability. Unlike traditional centrifuges, which are suited to enriching gases with a high molecular mass, ASP Technology is highly suited to of enriching gases with a low molecular mass such as silane (SiH4).
Quantum computers are expected to be thousands or millions of times more powerful than the most advanced of today’s conventional computers, opening new frontiers and opportunities in many industries, including medicine, artificial intelligence, cybersecurity, global logistics and global financial systems.
Quantum Enrichment Technology for Uranium Enrichment
We believe our Quantum Enrichment technology is capable of enriching Uranium, which we may be able to commercialize as a nuclear fuel component for use in the new generation of HALEU-fueled small modular reactors that are now under development for commercial and government uses.
Uranium is a naturally occurring element and is mined from deposits located in Kazakhstan, Canada, Australia, and several other countries including the United States. According to the World Nuclear Association (“WNA”), there are adequate measured resources of natural uranium to fuel nuclear power at current usage rates for about 90 years. In its natural state, uranium is principally comprised of two isotopes: uranium-235 (“U-235”) and uranium-238 (“U-238”). The concentration of U-235 in natural uranium is only 0.711% by weight. Most commercial nuclear power reactors require LEU fuel with a U-235 concentration greater than natural uranium and up to 5% by weight. Future reactor designs currently under development will likely require higher U-235 concentration levels of up to 20%. Uranium enrichment is the process by which the concentration of U-235 is increased (see discussion on HALEU demand below).
Separative work units(“SWU”) is a standard unit of measurement that represents the effort required to transform a given amount of natural uranium into two components: enriched uranium having a higher percentage of U-235 and depleted uranium having a lower percentage of U-235. The SWU contained in LEU is calculated using an industry standard formula based on the physics of enrichment. The amount of enrichment deemed to be contained in LEU under this formula is commonly referred to as its SWU component and the quantity of natural uranium deemed to be contained in LEU under this formula is referred to as its uranium or “feed” component. Currently, it is fairly common practice to purchase both the SWU and uranium components of LEU from the enrichment company. Therefore, LEU prices typically consist of three components: SWU, Conversion and uranium ore concentrate.
The following outlines the steps for converting natural uranium into LEU fuel, commonly known as the nuclear fuel cycle:
| · | Mining and Milling. Natural, or unenriched, uranium is removed from the earth in the form of ore and then crushed and concentrated. |
|
|
|
| · | Conversion. Uranium ore concentrates (“UO”) are combined with fluorine gas to produce uranium hexafluoride (“UF”), a solid at room temperature and a gas when heated. UF is shipped to an enrichment plant. |
|
|
|
| · | Enrichment. UF is enriched in a process that increases the concentration of the U isotope in the UF from its natural state of 0.711% up to 5%, or LEU, which is usable as a fuel for current light water commercial nuclear power reactors. Future commercial reactor designs may use uranium enriched up to 20% U-235, or HALEU. |
|
|
|
| · | Fuel Fabrication. LEU is then converted to uranium oxide and formed into small ceramic pellets by fabricators. The pellets are loaded into metal tubes that form fuel assemblies, which are shipped to nuclear power plants. As the advanced reactor market develops, HALEU may be converted to uranium oxide, metal, chloride or fluoride salts, or other forms and loaded into a variety of fuel assembly types optimized for the specific reactor design. |
|
|
|
| · | Nuclear Power Plant. The fuel assemblies are loaded into nuclear reactors to create energy from a controlled chain reaction. Nuclear power plants generate approximately 20% of U.S. electricity and 10% of the world’s electricity. |
|
|
|
| · | Used Fuel Storage. After the nuclear fuel has been in a reactor for several years, its efficiency is reduced and the assembly is removed from the reactor’s core. The used fuel is warm and radioactive and is kept in a deep pool of water for several years. Many utilities have elected to then move the used fuel into steel or concrete and steel casks for interim storage. |
The World is Transitioning to Newer Smaller Reactors
As the world transitions to a decarbonized electric grid, society is gradually decreasing its reliance on fossil fuels and increasing its reliance on “clean energy”. There appears to be bipartisan support for the growth of nuclear energy and the Biden Administration has identified carbon-free nuclear power as an essential part of achieving a net-zero CO2 economy by 2050. Nuclear power, through the operating light water reactor fleet and the deployment of advanced reactors, is poised to be an increasing contributor to carbon free energy in the U.S. and internationally. The United States leads the world in technology innovation with more developers of advanced reactors than any other country.
| 18 |
| Table of Contents |
Small modular reactors (SMRs) are advanced nuclear reactors that have a power capacity of up to 300 MW(e) per unit, which is about one-third of the generating capacity of traditional nuclear power reactors. SMRs, which can produce a large amount of low-carbon electricity, are:
| · | Small — physically a fraction of the size of a conventional nuclear power reactor. |
|
|
|
| · | Modular — making it possible for systems and components to be factory-assembled and transported as a unit to a location for installation. |
|
|
|
| · | Reactors — harnessing nuclear fission to generate heat to produce energy. |
Many of the benefits of SMRs are inherently linked to the nature of their design — small and modular. Given their smaller footprint, SMRs can be sited on locations not suitable for larger nuclear power plants. Prefabricated units of SMRs can be manufactured and then shipped and installed on site, making them more affordable to build than large power reactors, which are often custom designed for a particular location, sometimes leading to construction delays. SMRs offer savings in cost and construction time, and they can be deployed incrementally to match increasing energy demand.
In comparison to existing reactors, proposed SMR designs are generally simpler, and the safety concept for SMRs often relies more on passive systems and inherent safety characteristics of the reactor, such as low power and operating pressure. This means that in such cases no human intervention or external power or force is required to shut down systems, because passive systems rely on physical phenomena, such as natural circulation, convection, gravity and self-pressurization. These increased safety margins, in some cases, eliminate or significantly lower the potential for unsafe releases of radioactivity to the environment and the public in case of an accident.
SMRs have reduced fuel requirements. Power plants based on SMRs may require less frequent refueling, every 3 to 7 years, in comparison to between 1 and 2 years for conventional plants. Some SMRs are designed to operate for up to 30 years without refueling. SMRs are under construction or in the licensing stage in Argentina, Canada, China, Russia, South Korea and the United States of America.
Within the last five years significant legislation supporting the development and deployment of advanced reactors has been enacted: the Nuclear Innovation and Modernization Act, the Nuclear Energy Innovation and Capabilities Act, the Energy Act of 2020 and the Infrastructure Investment and Jobs Act. In addition, Congress established and funded the Advanced Reactor Demonstration Program which now supports two advanced reactor demonstrations to be deployed within seven years and eight other advanced reactor projects.
SMRs will require a different grade of enriched Uranium
Many advanced reactors, including the majority of the Advanced Reactor Demonstration Program awardees, will require High Assay Low Enriched Uranium (HALEU), and fuel forms very different from those manufactured for the current Light Water Reactors (LWRs). For example, the current generation of LWRs uses fuel enriched to less than 5% uranium-235. In contrast, many advanced non-LWR designs require enrichments between 5% and 20% with most above 10%.
Currently it is not possible to purchase HALEU between 10% and 20% from a commercial enricher in the United States. In the U.S., the infrastructure for the front-end of the fuel cycle for the utilization of low enriched uranium up to 5% U-235 is well defined. The U.S. has mining, conversion, enrichment, fabrication, and transportation capability. However, the infrastructure for producing and utilizing HALEU, in particular enrichments above 10%, is not established in the U.S. The mining and conversion infrastructure are common to all enrichment levels.
In 2020, the Department of Energy (DOE) selected two companies for awards under the Advanced Reactor Demonstration Program (ARDP) Pathway 1: Advanced Reactor Demonstrations. Both reactor designs require HALEU and can be operational in about seven years. Today, it is estimated that the companies selected for the demonstration pathway will require HALEU for their reactors beginning in 2024 to support fuel fabrication ahead of reactor startup. In addition, one of the companies under Pathway 2: Risk Reduction for Future Demonstrations will require HALEU in the 2024-2025 timeframe and other companies in Pathway 2 and 3 of the ARDP will also require HALEU. Privately funded companies are also working to deploy HALEU fueled reactors by the mid-2020s.
The Nuclear Energy Institute (NEI) believes that it is virtually impossible for HALEU to be provided to these companies in the needed quantities and timeframes from DOE inventories or commercial enrichers located in the U.S or Western Europe. Therefore, acquiring HALEU from other international suppliers will be required in the near term to support the larger goal of deploying advanced reactors in the U.S. in a timely manner. Deploying these reactors before 2030 will support climate goals and position the U.S. to be a strong exporter of advanced reactor technology. Per the recent NEI white paper, a robust domestic HALEU infrastructure is necessary to support both the domestic deployment of advanced reactors and the export of U.S. advanced reactor technologies requiring HALEU.
In a letter to the DOE captioned “Updated Need for High-Assay Low Enriched Uranium” dated December 20, 2021, the NEI provided an estimate of what U.S. HALEU demand may be during the next 15 years by companies denoted A to J:
| 19 |
| Table of Contents |
Estimated Annual Requirements for High Assay Low Enriched Uranium to 2035 (MTU/yr)
Company |
| A |
|
| B |
|
| C |
|
| D |
|
| E |
|
| F |
|
| G |
|
| H |
|
| I |
|
| J |
|
| Total |
|
| Cumulative |
| ||||||||||||
Year |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||||||||
2022 |
|
| 0.1 |
|
|
| 0.4 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 0.2 |
|
|
|
|
|
| 1.1 |
|
|
| 0.0 |
|
|
| 1.8 |
|
|
| 1.8 |
| |||||
2023 |
|
| 0.1 |
|
|
| 3.1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 4.4 |
|
|
| 0.1 |
|
|
| 7.7 |
|
|
| 9.5 |
| |||||
2024 |
|
| 1.0 |
|
|
| 5.6 |
|
|
| 0.2 |
|
|
| 3.0 |
|
|
|
|
|
|
|
|
| 1.5 |
|
|
|
|
|
| 6.6 |
|
|
| 0.1 |
|
|
| 18.0 |
|
|
| 27.5 |
| |||
2025 |
|
| 1.0 |
|
|
| 3.8 |
|
|
| 0.4 |
|
|
| 3.0 |
|
|
|
|
|
| 5.0 |
|
|
|
|
|
|
|
|
|
| 11.0 |
|
|
| 1.6 |
|
|
| 25.8 |
|
|
| 53.3 |
| ||
2026 |
|
| 1.0 |
|
|
| 15.1 |
|
|
|
|
|
|
| 4.9 |
|
|
|
|
|
| 10.0 |
|
|
| 2.0 |
|
|
| 24.2 |
|
|
| 13.2 |
|
|
| 1.7 |
|
|
| 72.1 |
|
|
| 125.4 |
| |
2027 |
|
| 1.0 |
|
|
| 26.5 |
|
|
|
|
|
|
| 7.9 |
|
|
|
|
|
|
|
|
|
| 4.0 |
|
|
| 24.2 |
|
|
| 13.2 |
|
|
| 1.9 |
|
|
| 78.7 |
|
|
| 204.1 |
| |
2028 |
|
| 1.0 |
|
|
| 37.8 |
|
|
|
|
|
|
| 16.6 |
|
|
|
|
|
| 13.0 |
|
|
| 23.0 |
|
|
| 24.2 |
|
|
| 13.2 |
|
|
| 2.0 |
|
|
| 130.8 |
|
|
| 334.9 |
| |
2029 |
|
| 1.0 |
|
|
| 26.3 |
|
|
| 1.8 |
|
|
| 30.5 |
|
|
| 17.0 |
|
|
| 18.0 |
|
|
| 14.0 |
|
|
| 24.2 |
|
|
| 16.5 |
|
|
| 2.4 |
|
|
| 151.7 |
|
|
| 486.6 |
|
2030 |
|
| 1.0 |
|
|
| 34.4 |
|
|
| 1.8 |
|
|
| 40.4 |
|
|
| 46.0 |
|
|
| 18.0 |
|
|
| 30.0 |
|
|
| 24.2 |
|
|
| 16.5 |
|
|
| 2.7 |
|
|
| 215.0 |
|
|
| 701.6 |
|
2031 |
|
| 23.0 |
|
|
| 42.5 |
|
|
| 6.2 |
|
|
| 53.0 |
|
|
| 29.0 |
|
|
| 22.0 |
|
|
| 33.0 |
|
|
| 24.2 |
|
|
| 16.5 |
|
|
| 2.9 |
|
|
| 252.3 |
|
|
| 954.0 |
|
2032 |
|
| 35.0 |
|
|
| 52.9 |
|
|
| 12.5 |
|
|
| 67.6 |
|
|
| 46.0 |
|
|
| 40.0 |
|
|
| 50.0 |
|
|
| 48.4 |
|
|
| 19.8 |
|
|
| 3.1 |
|
|
| 375.3 |
|
|
| 1329.2 |
|
2033 |
|
| 47.0 |
|
|
| 63.5 |
|
|
| 32.2 |
|
|
| 82.1 |
|
|
| 46.0 |
|
|
| 32.0 |
|
|
| 80.0 |
|
|
| 48.4 |
|
|
| 19.8 |
|
|
| 3.2 |
|
|
| 454.2 |
|
|
| 1783.4 |
|
2034 |
|
| 58.0 |
|
|
| 76.1 |
|
|
| 62.4 |
|
|
| 96.7 |
|
|
| 46.0 |
|
|
| 36.0 |
|
|
| 80.0 |
|
|
| 48.4 |
|
|
| 19.8 |
|
|
| 3.7 |
|
|
| 527.1 |
|
|
| 2310.5 |
|
2035 |
|
| 70.0 |
|
|
| 90.9 |
|
|
| 96. |
|
|
| 112.4 |
|
|
| 91.0 |
|
|
| 29.0 |
|
|
| 50.0 |
|
|
| 48.4 |
|
|
| 22.0 |
|
|
| 4.1 |
|
|
| 613.8 |
|
|
| 2924.3 |
|
Notes:
| · | The material needs listed above are in metric tons of uranium per year and are a small amount compared to the approximately 2000 MTU used annually by the existing fleet of reactors. |
|
|
|
| · | The material needs listed above include enrichments between 10.9% and 19.75% U-235. |
|
|
|
| · | The year the material is needed is for fuel fabrication. Insertion in the reactor and reactor operations will occur in a later year. |
|
|
|
| · | The material needs that are less than 1 MTU/year are for irradiation samples, lead test rods and lead test fuel assemblies. |
|
|
|
| · | The material needs represent a few scenarios |
| o | The deployment of an advanced fuel design for the existing fleet of light-water reactors. |
|
|
|
| o | The deployment of multiple reactors of the same design that will not require refueling for many years. |
|
|
|
| o | The deployment of reactors that have annual refueling requirements. |
| · | These reactors include a range of sizes from a few Megawatt electric to 100s of Megawatt electric. |
|
|
|
| · | The data above does not include utilities that are considering enrichment between 5% and 10%. |
Quantum Enrichment Technology is ideally suited to the production of HALEU
We believe that we are in a very different position to many of the entrenched domestic and international enrichers. Our innovative isotope enrichment process has a number of advantages over traditional gas centrifuges and other novel approaches currently being explored by other companies: cheaper in Capex, faster in construction, more flexible in design and location.
We estimate that the capital cost of constructing Quantum Enrichment plant for uranium enrichment is approximately 75% cheaper than that of a traditional gas centrifuge enrichment facility. Our manufacturing plants are modular, so our construction time is likely faster and more flexible than competing technologies. Our enrichment facilities are smaller than traditional gas centrifuges which means we can place them near fuel fabrication facilities for enhanced security of production and transportation. Our operating costs of enriching uranium to 15.5% - 19.75% U-235 should be comparable to or cheaper than costs for other methods of uranium enrichment.
| 20 |
| Table of Contents |
The table below represents management’s estimated comparison of the Quantum Enrichment process with a traditional gas centrifuge.
|
| Quantum Enrichment Plant |
| Gas Centrifuge |
Separation mechanism |
| Enhanced resonant multiphoton ionization |
| Differential diffusion |
Capital Cost per plant |
| <$100 million |
| >$800 million |
Energy use (kWh) per SWU |
| <40 |
| 50-240 |
Construction time |
| 2-3 years |
| 2-3 years |
Levelized cost per SWU* |
| <$50 |
| $140 |
___________
* | for enrichment from 0.71% U235 to 5% U235 |
We are currently constructing a Ytterbium-176 enrichment facility using the Quantum Enrichment technology in Pretoria, South Africa. We received a manufacturing permit for this facility from the South African Department of Mineral Resources and Energy (DMRE) during 3Q 2023. The construction of this plant will provide us with valuable experience in the construction of Quantum Enrichment facilities in the future. Many of the control systems, compressors, lasers and hardware used in a uranium enrichment facility would be similar to parts used in this ytterbium-176 enrichment facility.
We expect the construction of a Uranium Enrichment facility would take approximately 30 months and the production volume would gradually ramp up to the final capacity of 20 metric tons per year. Importantly, subject to licensure, we believe we can produce commercial quantities of HALEU by 2027 that would satisfy the anticipated demand from all the advanced reactor currently in development. We believe that we can supply HALEU at a price lower than the HALEU currently imported from international enrichers and considerably lower than any potential domestic supply that may evolve.
Intellectual Property
Our business will depend on the proprietary ASP technology that was developed by and originally licensed to us from Klydon. To date, we have relied exclusively on trade secrets and other intellectual property laws, non-disclosure agreements with our respective employees, consultants, vendors, potential customers and other relevant persons and other measures to protect our intellectual property, and intend to continue to rely on these and other means. As we intend to transition into the commercialization of isotopes, we envision our intellectual property and its security becoming more vital to our future. Pursuing patent protection remains part of the intellectual property protection philosophy and strategy and the advisability of establishing provisional patent rights is continuously assessed on a case-by-case basis in respect of both conceptual aspects and the specific applications thereof. Such assessments are made in consultation with regulatory bodies and with due consideration to the prospects of successfully obtaining patent protection in light of any disclosure constraints that are imposed by such bodies.
Regulatory Environment
We are subject to a variety of laws and regulations, including but not limited to those of the United States and South Africa, that impose regulatory systems that govern many aspects of our operations, including our research and development activities involving the enrichment of isotopes in South Africa. In addition, these jurisdictions impose trade controls requirements that restrict trade to comply with applicable export controls and economic sanctions laws and requirements, and legal requirements that are intended to curtail bribery and corruption.
There are a number of regulators and treaties that govern and control our business and industry. The two principal ones that control and regulate the manufacturing of isotopes at our isotope enrichment facility in South Africa are the International Atomic Energy Agency (IAEA) and the Nuclear Non-Proliferation Treaty (NPT).
The IAEA is an international organization that seeks to promote the peaceful use of nuclear energy, and to inhibit its use for any military purpose, including nuclear weapons. The IAEA was established as an autonomous organization on 29 July 1957. Though established independently of the United Nations through its own international treaty, the IAEA Statute, the IAEA reports to both the United Nations General Assembly and Security Council. The IAEA statute currently has 173 member states, including South Africa.
The IAEA is authorized to conclude agreements with member states, in terms of which agreements the agency would perform certain functions and the relevant member states would be placed under certain obligations. The IAEA has concluded an extensive suite of agreements with South Africa. These agreements can be viewed on the website of the IAEA (https://www.iaea.org/resources/legal/country-factsheets) and include agreements that govern the physical protection of nuclear material, the notification of nuclear accidents, assistance in the case of nuclear accidents, nuclear safety, civil liability, and technical cooperation.
| 21 |
| Table of Contents |
The Treaty on the Non-Proliferation of Nuclear Weapons, commonly known as the Non-Proliferation Treaty or NPT, is an international treaty whose objective is to prevent the spread of nuclear weapons and weapons technology, to promote cooperation in the peaceful uses of nuclear energy, and to further the goal of achieving nuclear disarmament and general and complete disarmament. Our South African subsidiary is registered with the South African Council for the Non-Proliferation of Weapons of Mass Destruction in terms of the Non-Proliferation of Weapons of Mass Destruction Act, 1993. Our registration certificate is valid until September 3, 2023. Representatives from the South African Council for the Non-Proliferation of Weapons of Mass Destruction regularly inspect our facility and conduct tests to monitor the activities that are taking place at our facilities.
In South Africa, government Notice 493 relates to nuclear-related dual-use equipment, materials and software and related technologies which can be used in their entirety or in part for the separation of uranium isotopes. ASP is classified as a dual use technology under the protocols of the IAEA and, as such, is subject to the controls that are implemented under these protocols. These controls comprise requirements that include:
| · | membership of the IAEA and adherence to its protocols; |
|
|
|
| · | membership of the Nuclear Suppliers Group (NSG) and adherence to its protocols; |
|
|
|
| · | agreement to an “additional protocol” in light of uranium enrichment capabilities; |
|
|
|
| · | local laws that require permits for possession, operation and commercialization and regular reporting; |
|
|
|
| · | ad hoc inspections by the IAEA on 24 hour and in some cases 2 hours pre-warning; |
|
|
|
| · | requirement for proposed patent applications to be approved at ministerial level; and |
|
|
|
| · | cross-border technology transfer to be handled by the respective governments and approved by IAEA. |
These regulations place strict limitations on what we can and cannot do. Security measures at our production facility and our offices are stringent. Access to our manufacturing plant is highly controlled. All employees and all visitors to the manufacturing plant are pre-screened by the South African Council for the Non-Proliferation of Weapons of Mass Destruction before being allowed employment or entry into the facility. Some of our suppliers also need to be registered with the South African Council for the Non-Proliferation of Weapons of Mass Destruction. Many of our computer systems are not connected to the external internet and confidential information is secured at a controlled location.
Currently, the production, distribution or sale of Mo-100 or Zn-68 is not regulated by a healthcare regulator such as the Food and Drug Administration (FDA) in the USA, Health Canada in Canada, the European Medicines Agency in Europe and similar regulators in other countries. However, products that are produced from Mo-100 or Zn-68 (such as Mo-99 and Tc-99m in a linear accelerator or cyclotron and Ga-67 in a cyclotron) are regulated by healthcare regulators and our customers are required to operate under the licensure of these healthcare regulators. Currently, the production and use of Tc-99m from Mo-100 in a cyclotron is only approved in one country (Canada).
Some of our future isotopes may also be regulated by healthcare regulators such as the Food and Drug Administration (FDA) in the USA, Health Canada in Canada, the European Medicines Agency in Europe and similar regulators in other countries.
U.S. laws restrict the ability of U.S. companies, U.S. citizens and U.S. permanent residents, or U.S. persons, from involvement in certain types of transactions with countries, businesses and individuals that have been targeted by U.S. economic sanctions. For example, U.S. persons are precluded from undertaking virtually any activity of any kind on the part of any U.S. person with regard to any potential or actual transactions involving Cuba, Iran and Sudan without the prior approval of the U.S. Department of Treasury’s Office of Foreign Assets Control, or OFAC. OFAC also administers U.S. sanctions against a lengthy list of entities and individuals, wherever they may be located, that the United States considers to be closely associated with these sanctioned countries or that are considered terrorists or traffickers in either narcotics or weapons of mass destruction. Furthermore, U.S. economic sanctions forbid U.S. persons from circumventing direct U.S. restrictions or from facilitating transactions by non-U.S. persons if those activities are forbidden to U.S. persons. Penalties for violating provisions such as these can include significant civil and criminal fines, imprisonment and loss of tax credits or export privileges.
The Foreign Corrupt Practices Act of 1977, or the FCPA, as amended by the Omnibus Trade and Competitiveness Act of 1988 and the International Anti-Bribery and Fair Competition Act of 1998, makes it a criminal offense for a U.S. corporation or other U.S. domestic concern to make payments, gifts or give anything of value directly or indirectly to foreign officials for the purpose of obtaining or retaining business, or to obtain any other unfair or improper advantage. In addition, the FCPA imposes accounting standards and requirements on publicly traded U.S. corporations and their foreign affiliates, which are intended to prevent the diversion of corporate funds to the payment of bribes and other improper payments, and to prevent the establishment of “off books” slush funds from which such improper payments can be made. We are also subject to laws and regulations covering subject matter similar to that of the FCPA that have been enacted by countries outside of the United States. For example, the Convention on Combating Bribery of Foreign Public Officials in International Business Transactions was signed by the members of the Organization for Economic Cooperation and Development and certain other countries in December 1997. The Convention requires each signatory to enact legislation that prohibits local persons and firms from making payments to foreign officials for the purpose of obtaining business or securing other unfair advantages from foreign governments. Failure to comply with these laws could subject us to, among other things, penalties and legal expenses, which could harm our reputation and have a material adverse effect on our business, financial condition and results of operations.
| 22 |
| Table of Contents |
Compliance with the myriad of export control laws of the various jurisdictions in which we do business is a challenge for any company involved in export activities within the nuclear and defense end markets. We have compliance systems in our U.S. and non-U.S. subsidiaries to identify those products and technologies that are subject to export control regulatory restrictions and, where required, we obtain authorization from relevant regulatory authorities for sales to foreign buyers or for technology transfers to foreign consultants, companies, universities or foreign national employees. We also have a compliance system that is intended to proactively address potential compliance issues including those related to export control, trade sanctions and embargoes, as well as anti-bribery situations, and we are implementing this through such mechanisms as training, formalizing contracting processes, performing diligence on agents and continuing to improve our record-keeping and auditing practices with respect to third-party relationships and otherwise. Thus far, as part of our compliance system, for instance, we have developed a Code of Ethics and Conduct that informs all of our employees of their compliance obligations. Furthermore, we have developed an ethics and conduct training program that all of our employees are required to undertake, as well as other targeted compliance training relevant to their position, such as specific FCPA training for all of our worldwide controllers. Violations of any of the various U.S. or non-U.S. export control laws can result in significant civil or criminal penalties, or even loss of export privileges, as mentioned above. We recognize that an effective compliance program can help protect the reputation and relationship of a regulated company with the regulatory agencies administering these laws and regulations. In the United States, each of the regulatory agencies administering these laws and regulations has a voluntary disclosure program that offers the possibility of significantly reduced penalties, if any are applicable, and we intend to use these programs as part of our overall compliance program, as necessary.
Employees
As of December 31, 2023, we employed 76 people on a full-time basis. Of the total employees, 6 employees are in research and development, 33 employees are in engineering, construction and manufacturing, 20 employees are in plant operations and 17 employees are in general management. None of our employees are subject to collective bargaining agreements. We consider our relationship with our employees to be good.
Facilities
We lease five facilities in Pretoria, South Africa for production, research and development and offices. One lease is under automatic monthly extensions, another lease which expired on March 31, 2024 is now under automatic monthly extensions and the other three leases have terms that expire between March 30, 2026 and December 31, 2030. We believe that our existing facilities are adequate to meet our current needs.
Legal Proceedings
We are currently not a party to any material legal proceedings.
Item 1A. Risk Factors
Investing in our common stock involves a high degree of risk. You should carefully consider the risk factors below together with the information contained elsewhere in this Annual Report on Form 10-K, including Part II, Item 8 “Financial Statements and Supplementary Data” and Part II, Item 7, “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” and in our other public filings in evaluating our business. Before you decide to invest in our common stock, you should consider carefully the risks described below, together with the other information contained in this Annual Report, including our financial statements and the related notes. We believe the risks described below are the risks that are material to us as of the date of this Annual Report. If any of the following risks actually occur, our business, financial condition, results of operations and future growth prospects could be materially and adversely affected. In these circumstances, the market price of our common stock could decline, and our stockholders may lose all or part of their investment.
| 23 |
| Table of Contents |
Summary of the Material and Other Risks Associated with Our Business
Our business is subject to numerous material and other risks and uncertainties that you should be aware of in evaluating our business. These risks include, but are not limited to, the following:
| · | We have incurred significant net losses since inception, and we expect to continue to incur significant net losses for the foreseeable future. |
|
|
|
| · | We have a limited operating history, which may make it difficult to evaluate our prospects and likelihood of success. |
|
|
|
| · | Our business is tied directly to the nuclear medicine industry and depends on our ability to successfully introduce our Mo-100 and other medical isotopes to changing technology and a changing medical practice landscape. |
|
|
|
| · | Our business is dependent on our ability to recognize the anticipated benefits of acquisitions, including our acquisition of assets of Molybdos (Pty) Limited in the “business rescue” auction, the assets and intellectual property we acquired from Klydon Proprietary Ltd, and our investment in PET Labs Pharmaceuticals; |
|
|
|
| · | We currently have no sales attributable to isotopes, but we expect to be heavily dependent on a few large customers to generate a majority of our revenues from sales of our future isotopes. Our operating results could be adversely affected by a reduction in business with our future significant customers. |
|
|
|
| · | We are still conducting research and development efforts for isotopes such as Mo-100, Zinc-68, Silicon-28, Xenon-129/136, Germanium-70/72/74 and Chlorine-37 using the ASP technology. If we are unable to advance our future isotopes in development, obtain applicable regulatory approval and ultimately commercialize our future isotopes, or experience significant delays in doing so, our business will be materially harmed. |
|
|
|
| · | We are awaiting the approvals necessary to conduct early research and development efforts for isotopes such as Uranium-235 utilizing the Quantum Enrichment process. The necessary approvals may take a significant amount of time and may never fail to materialize. As a result, we will not be able to enter into the nuclear energy space utilizing our technology. |
|
|
|
| · | Obtaining and maintaining our patent protection depends on compliance with various procedures, document submissions, fee payments and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements. |
|
|
|
| · | Since our listing on the Nasdaq Capital Market in November 2022, there has been only a limited prior public market for our Common Stock, the stock price of our Common Stock may be volatile or may decline regardless of our operating performance and you may not be able to resell your shares quickly or at the market price if trading in shares of our common stock is not active. |
|
|
|
| · | If we fail to maintain an effective system of internal control over financial reporting, we may not be able to accurately report our financial results or prevent fraud. As a result, stockholders could lose confidence in our financial and other public reporting, which would harm our business and the trading price of our Common Stock. |
|
|
|
| · | Our independent registered public accounting firm’s report contains an explanatory paragraph that expresses substantial doubt about our ability to continue as a “going concern.” |
The material and other risks summarized above should be read together with the text of the full risk factors below and in the other information set forth in this Annual Report, including our consolidated financial statements and the related notes, as well as in other documents that we file with the SEC. If any such material and other risks and uncertainties actually occur, our business, prospects, financial condition and results of operations could be materially and adversely affected. The risks summarized above or described in full below are not the only risks that we face. Additional risks and uncertainties not currently known to us, or that we currently deem to be immaterial may also materially adversely affect our business, prospects, financial condition and results of operations.
| 24 |
| Table of Contents |
Risks Related to Our Limited Operating History, Financial Position and Need for Additional Capital
We have a very limited operating history, and we have incurred losses since our inception and anticipate that we will continue to incur significant losses for the foreseeable future. We may never generate any revenue attributable to sales of enriched isotopes or become profitable or, if we achieve profitability, we may not be able to sustain it.
We were incorporated in September 2021, and we have a very limited operating history upon which you can evaluate our business and prospects. Our operations to date have been primarily focused on acquiring the assets of Molybdos (after participating in and being declared the winner of a competitive auction process under Section 45 of the South Africa Consumer Protection Act, 2008 for ZAR 11,000,000, which at the then current exchange rate was approximately $734,000) and in-licensing intellectual property rights related to the production of Molybdenum-100 (a non-radioactive isotope we believe may have applications primarily in the medical industry) and Uranium-235 (an isotope of uranium we believe may have application in the clean, efficient and carbon-free energy industry) using the ASP technology, organizing and staffing our company, research and development activities, business planning, raising capital, and providing general and administrative support for these operations. In July 2022, we acquired assets comprising a dormant Silicon-28 aerodynamic separation processing plant from Klydon. We have not yet built a manufacturing plant that is producing commercial quantities of isotopes or even demonstrated the ability to produce commercial quantities of isotopes using the ASP technology or quantum enrichment technology. We have not yet demonstrated an ability to overcome many of the risks and uncertainties frequently encountered by companies in the medical, technology and energy industries, including an ability to obtain applicable regulatory approvals, manufacture any isotopes at commercial scale, or conduct sales and marketing activities necessary for successful isotope commercialization. In addition, we have not yet sought any regulatory approval that may be necessary for application of Mo-100 that we may produce using the ASP process in the medical industry or the production of U-235 that we may produce using quantum enrichment. Consequently, any predictions about our future performance may not be as accurate as they would be if we had a history of successfully developing and commercializing isotopes.
Investment in isotope enrichment technology is highly speculative because it entails substantial upfront capital expenditures and significant risk that any potential isotopes will fail to demonstrate adequate utility or effectiveness in the targeted application (or for medical indications, an acceptable safety profile), gain regulatory approval, if applicable, and become commercially viable. We have no products approved for commercial sale and have not generated any revenue to date attributable to isotopes (and only limited revenues attributable to PET Labs), and we continue to incur significant research and development and other expenses related to our ongoing operations. As a result, we are not profitable and have incurred losses since our inception in September 2021. For the years ended December 31, 2023 and 2022, we reported a net loss of $16.3 million and $4.9 million, respectively. As of December 31, 2023, we had an accumulated deficit of $23.8 million.
We expect to continue to incur significant losses for the foreseeable future, and we expect these losses to increase as we:
| · | continue to invest in our research and development activities; |
|
|
|
| · | seek applicable regulatory approvals for any future isotopes that we may successfully develop; |
|
|
|
| · | experience any delays or encounter any issues with any of the above, including but not limited to failed research and development activities, safety issues, or other regulatory challenges; |
|
|
|
| · | hire additional engineering and production personnel and build our internal resources, including those related to audit, patent, other legal, regulatory and tax-related services associated with maintaining compliance with exchange listing and SEC requirements, director and officer insurance premiums and investor and public relations costs; |
|
|
|
| · | obtain, expand, maintain, enforce and protect our intellectual property portfolio; |
|
|
|
| · | establish a sales, marketing and distribution infrastructure and establish manufacturing capabilities, whether alone or with third parties, to commercialize future isotopes (assuming receipt of applicable regulatory approvals), if any; and |
|
|
|
| · | operate as a public company. |
We expect limited commercial activity for our isotopes in the United States during the next two to three years and we anticipate that most of our initial revenues from future sales of our Mo-100 will be derived from countries in Asia and EMEA (Europe, Middle East and Africa). To become and remain profitable, we must succeed in developing and eventually commercializing enriched isotopes that generate significant revenue. This will require us to be successful in a range of challenging activities, including completing research and development activities relating to our ASP technology, obtaining applicable regulatory approval for future isotopes, if any, and manufacturing, marketing and selling any future isotopes (assuming receipt of applicable regulatory approvals). We are only in the preliminary stages of most of these activities. We may never succeed in these activities and, even if we do, may never generate revenues that are significant enough to achieve profitability. Because of the numerous risks and uncertainties associated with chemical isotopes separation, we are unable to accurately predict the timing or amount of increased expenses or when, or if, we will be able to achieve profitability. Even if we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis. Our failure to become and remain profitable would depress the value of our company and could impair our ability to raise capital, expand our business, maintain our research and development efforts, diversify our future isotopes or even continue our operations. A decline in the value of our company could also cause you to lose all or part of your investment.
| 25 |
| Table of Contents |
Our future prospects are tied directly to the end markets that use our isotopes including the diagnostic medical imaging industry and depend on our ability to successfully introduce our isotopes and adapt to a changing technology and medical practice landscape.
The field of diagnostic medical imaging is dynamic, with new products, including equipment, software and products, continually being developed and existing products continually being refined. New hardware (scanners), software or agents in a given diagnostic modality may be developed that provide benefits superior to the then-dominant hardware, software and agents in that modality, resulting in commercial displacement of the existing radiotracers. For example, alternate scanners and radiotracers could be introduced. Similarly, changing perceptions about comparative efficacy and safety, as well as changing availability of supply may favor one agent over another or one modality over another. In addition, new or revised appropriate use criteria developed by professional societies, to assist physicians and other health care providers in making appropriate imaging decisions for specific clinical conditions, can and have reduced the frequency of and demand for certain imaging modalities and imaging agents. Technological obsolescence in any of the medical imaging products that would use the Mo-100 that we plan to manufacture could have a material adverse effect on our business, results of operations, financial condition and cash flows.
We may not realize the anticipated benefits of previous acquisitions.
The success of the company will depend in large part on the success of our management in integrating the acquired assets into the company. In October 2021, our subsidiary in South Africa acquired the assets of Molybdos after participating in and being declared the winner of a competitive auction process under Section 45 of the South Africa Consumer Protection Act, 2008 for ZAR 11,000,000 (which at the then current exchange rate was approximately $734,000), plus value added tax (VAT) levied by the government of South Africa at the rate of 15% and auctioneers’ commission at the rate of 10%. In July 2022, we acquired assets comprising a dormant Silicon-28 aerodynamic separation processing plant from Klydon located in Pretoria, South Africa for ZAR 6,000,000 (which at the then current exchange rate was approximately $364,000). In addition, in April 2023, we perfected our interest under the Acknowledgement of Debt Agreement, under which we acquired specific intellectual property from Klydon. To date, we have completed the construction of one isotope enrichment facility, but we have not yet produced any commercial quantities of isotopes and we have not yet demonstrated the ability to produce any isotope in commercial quantities using ASP technology. We will not know whether the assets that we acquired will work according to our expectations until we have produced commercial quantities of isotopes at our enrichment facilities. Our failure to achieve the integration of the acquired assets into the company and to commercialize the assets could result in our failure to realize the anticipated benefits of those acquisitions and could impair our results of operations, profitability and financial results.
The acquisition of a controlling interest in Pet Labs Pharmaceuticals may fail to result in anticipated benefits but has involved significant investment of financial and other resources.
In October 2023, we entered into a Share Purchase Agreement with Nucleonics Imaging Proprietary Limited, a company incorporated in South Africa, to purchase 51% of the ordinary shares (the “initial shares”) in Nucleonics’ wholly-owned subsidiary, Pet Labs Pharmaceuticals Proprietary Limited, a company incorporated in South Africa and dedicated to nuclear medicine and the science of radiopharmaceutical production. We agreed to pay a total of $2,000,000 for the initial shares in two installments. The first installment of $500,000 was paid in November 2023. The remaining balance of $1,500,000 is due upon demand any time after October 31, 2024 and is expected to be paid in November 2024. In addition, we have an option to purchase the remaining 49% of the ordinary shares (the “option shares”). If we exercise our option to purchase the option shares (which option is exercisable until January 31, 2027, provided that the initial shares have been paid for in full), we have agreed to pay $2,200,000 for the option shares.
Acquisitions generally create risks such as (i) the need to integrate and manage the businesses and products acquired with our own business and products; (ii) additional demands on our resources, systems, procedures and controls; (iii) disruption of our ongoing business; (iv) potential unknown or unquantifiable liabilities associated with the target company; and (v) diversion of management’s attention from other business concerns. Moreover, this acquisition involves substantial investment of funds. This acquisition may not be successful in generating material revenue, income or other returns, and any resources we committed will not be available to us for other purposes. Our inability to take advantage of growth opportunities or address risks associated with this acquisition and investment may negatively affect our operating results. This acquisition may not result in its anticipated benefits, and we may not be able to properly integrate the business with our future products and operations or successfully combine personnel and cultures. Failure to do so could deprive us of the intended benefits of this acquisition.
| 26 |
| Table of Contents |
We currently have no sales attributable to enriched isotopes, but we expect to be heavily dependent on a few large customers to generate a majority of our revenues. Our operating results could be adversely affected by a reduction in business with our future significant customers.
We currently have no sales attributable to enriched isotopes. However, we expect to rely on a limited number of customers to purchase any isotopes that we produce using the ASP technology or quantum enrichment under long-term contracts. Our future key customers may stop ordering our isotopes at any time or may become bankrupt or otherwise unable to pay. The loss of any of our future key customers could result in lower revenues than we anticipate and could harm our business, financial condition or results of operations.
Our independent registered public accounting firm’s report contains an explanatory paragraph that expresses substantial doubt about our ability to continue as a “going concern.”
We generated an accumulated deficit totaling approximately $23.8 million through December 31, 2023. As of December 31, 2023, we had approximately $7.9 million in cash. Prior to our acquisition of 51% of PET Labs Pharmaceuticals, we had yet to generate any revenues, and we anticipate that our losses will continue for the foreseeable future. We cannot assure you that our plans to commercialize enriched isotopes that we may produce will be successful. These factors, among others, raise substantial doubt about our ability to continue as a going concern. The financial statements contained elsewhere in this report do not include any adjustments that might result from our inability to continue as a going concern. Unless we can begin to generate material revenue from production and sale of enriched isotopes or raise capital from equity offerings, we may not be able to remain in business. We cannot assure you that we will raise enough money or generate sufficient sales to meet our future working capital needs.
We will require substantial additional capital to finance our operations, which may not be available on acceptable terms, or at all. Failure to obtain this necessary capital when needed may force us to delay, limit or terminate certain of our product development efforts or other operations.
We expect our expenses to increase substantially in connection with our ongoing and planned activities, particularly as we continue our research and development activities, seek applicable regulatory approvals for any future isotopes that we may successfully develop, and expand our organization by hiring additional personnel. In addition, we expect to continue incurring significant costs associated with operating as a public company.
As of December 31, 2023, our cash was approximately $7.9 million. Subsequent to the end of 2023, in March 2024, QLE sold unsecured promissory notes for aggregate cash consideration of approximately $20.5 million. In addition, in April 2024, the Company received approximately $5.5 million from the issuance of 3,164,557 shares of common stock upon the exercise of warrants. We believe, based on our current operating plan, that the net proceeds from our IPO, private placements completed in March 2023, October 2023, promissory notes issued in March 2024, and the exercise of warrants in April 2024 together with our existing cash and cash equivalents, will not be sufficient to fund our operations for at least the next 12 months from the date the financial statements are issued. Therefore, we may need to seek additional funds through public or private equity or debt financings, third-party funding, marketing and distribution arrangements, as well as other collaborations, strategic alliances and licensing arrangements, or any combination of these approaches.
In any event, we will require substantial additional capital to support our business operations as we pursue additional research and development activities related to our ASP technology and seek applicable regulatory approval of our any future isotopes, and otherwise to support our continuing operations. In addition, we expect to incur significant commercialization expenses related to product sales, marketing, manufacturing and distribution (assuming receipt of applicable regulatory approvals for our future isotopes). Even if we believe we have sufficient capital for our current or future operating plans, we may seek additional capital if market conditions are favorable or if we have specific strategic considerations. Any additional capital raising efforts may divert our management from their day-to-day activities, which may adversely affect our ability to develop and commercialize our future isotopes (assuming receipt of applicable regulatory approvals).
Additionally, as a result of severely diminished liquidity and credit availability, increased interest rates, inflationary pressures, declines in consumer confidence, declines in economic growth, increases in unemployment rates and uncertainty about economic stability, the global credit and financial markets have experienced extreme volatility and disruptions, including severely diminished liquidity and credit availability. The financial markets and the global economy may also be adversely affected by the current or anticipated impact of military conflict. If the equity and credit markets deteriorate, it may make any necessary debt or equity financing more difficult, more costly or more dilutive. If we do not raise additional capital in sufficient amounts, we may be prevented from pursuing development and commercialization efforts, which will harm our business, operating results and prospects.
| 27 |
| Table of Contents |
We are subject to credit counterparty risk which could have a material adverse effect on our business, results of operations, financial condition and cash flows.
The Company maintains cash balances at many financial institutions in multiple geographies. While the majority of cash balances are currently held in USD at U.S. financial institutions, our cash balances at those institutions may exceed the Federal Deposit Insurance Corporation (“FDIC”) insurance limit of $250,000 per depositor, per insured bank for each account ownership category. Our non-US banking counterparties might not have protections offered to their customers that are considered standard in the U.S. and even if such deposit insurances do exist, there is no guarantee that the insurer will honor those insurance policies. Although the Company currently believes that the financial institutions with whom it does business, will be able to fulfill their commitments to the Company, there is no assurance that those institutions will be able to continue to do so. Any credit losses that may occur could have a material adverse effect on our business, results of operations, financial condition and cash flows.
Raising additional capital or acquiring or licensing assets by issuing equity or debt securities may cause dilution to our stockholders, and raising funds through lending and licensing arrangements may restrict our operations or require us to relinquish proprietary rights.
We may plan to seek additional capital through a combination of public and private equity offerings, debt financings, strategic partnerships and alliances and licensing arrangements. To the extent that we raise additional capital through the sale of equity or convertible debt securities, your ownership interest will be diluted, and the terms may include liquidation or other preferences that adversely affect your rights as a stockholder. The incurrence of indebtedness would result in increased fixed payment obligations and could involve certain restrictive covenants, such as limitations on our ability to incur additional debt, limitations on our ability to acquire or license intellectual property rights and other operating restrictions that could adversely impact our ability to conduct our business. If we raise additional capital through future collaborations, strategic alliances or third-party licensing arrangements, we may have to relinquish valuable rights to our intellectual property, future revenue streams, research programs or future isotopes, or grant licenses on terms that may not be favorable to us.
If we are unable to raise additional capital when needed, we may be required to delay, limit, reduce or terminate our product development or future commercialization efforts, or grant rights to develop and market our future isotopes (assuming receipt of applicable regulatory approvals for our future isotopes) that we would otherwise develop and market ourselves.
Risks Related to the Development and Commercialization of Our Future Isotopes
We are early in our research and development efforts for isotopes using the ASP technology and the quantum enrichment process. If we are unable to advance our future isotopes in development, obtain applicable regulatory approval and ultimately commercialize our future isotopes, or experience significant delays in doing so, our business will be materially harmed.
We are still conducting research and development efforts using ASP technology to produce a wide array of isotopes, and have not yet produced any isotope at commercial scale. It is possible that the research and development, proof-of-concept, construction of a plant and commercialization will take longer than anticipated due to unexpected delays.
We also plan to begin researching the enrichment of uranium, which is a chemical element we believe may have application in the clean, efficient and carbon-free energy industry, using quantum enrichment. Quantum enrichment has never been used to produce isotopes at a commercial scale and the research that has been conducted using this technique has never been published. The IAEA has never inspected any facility that leverages this technology and there is no proof that this technology has ever been used to enrich uranium. There are significant regulatory hurdles associated with enabling our research and development efforts to enter the nuclear energy market. Multiple regulatory agencies need to provide approvals to allow us to proceed with the research and development necessary to show proof of concept to the market. If we demonstrate proof of concept, we anticipate that there will be further approvals needed to expand to a larger footprint to support commercial demand. We may not ever obtain these approvals. If we are unable to advance our future isotopes in development, obtain applicable regulatory approval and ultimately commercialize our future isotopes (assuming receipt of applicable regulatory approvals), or experience significant delays in doing so, our business will be materially harmed.
Our ability to generate product revenues will depend heavily on the success of our research and development activities, receipt of applicable regulatory approvals, and eventual commercialization of our future isotopes (assuming receipt of applicable regulatory approvals and compliance with all applicable regulatory authorities).
The success of our business, including our ability to finance our company and generate any revenue in the future, will primarily depend on the successful development, regulatory approval and commercialization of our currently planned future isotopes, which may never occur.
We will have to be successful in a range of challenging activities, including completing research and development activities relating to our ASP technology, obtaining applicable regulatory approval for future isotopes, if any, and manufacturing, marketing and selling any future isotopes (assuming receipt of applicable regulatory approvals). We are only in the preliminary stages of most of these activities. If we are unable to succeed in these activities, we may not be able to generate sufficient revenue to continue our business.
| 28 |
| Table of Contents |
We rely on a limited number of suppliers to provide us components and a material interruption in supply could prevent or limit our ability to execute our strategic plan and development programs in the expected timeframe.
We depend upon a limited number of third-party suppliers for certain components required to construct the centrifuges and other equipment for the enrichment plants that are being constructed in South Africa. To date, we have been able to obtain the required components for our centrifuges without any significant delays or interruptions, except for certain delays related to COVID-19. If we lose any of these suppliers, we may be required to find and enter into supply arrangements with one or more replacement suppliers. Obtaining alternative sources of supply could involve significant delays and other costs, and these supply sources may not be available to us on reasonable terms or at all. Any disruption of supplies could delay completion of the enrichment plant in South Africa, which could adversely affect our ability to execute our strategic plan and development programs in the expected timeframe.
Development activities at our facility in South Africa could be disrupted for a variety of reasons, which could prevent us from completing our development activities.
A disruption in development activities at our facility in South Africa could have a material adverse effect on our business. Disruptions could occur for many reasons, including power outages, fire, natural disasters, weather, unplanned maintenance or other manufacturing problems, public health crises, disease, strikes or other labor unrest, transportation interruption, government regulation, political unrest or terrorism. Alternative facilities with sufficient capacity or capabilities may not be available, may cost substantially more or may take a significant time to start production, each of which could negatively affect our business and financial performance. South Africa struggles with limited electricity supply and regions of the country regularly undergo load-shedding, during which electricity is not available. This uncertain supply of electricity could impact our ability to operate and produce commercial products and could negatively affect the financial position of the Company.
Risks associated with the development of ASP technology for enrichment of isotopes could cause substantial delays in production of our future isotopes.
Prior to October 2021, as a company, we had no involvement with or control over the research and development of the ASP technology. We relied on Klydon to conduct such research and development in accordance with the applicable legal, regulatory and scientific standards. If the research and development processes or the results of the development programs associated with the ASP technology for development of isotopes prove to be unreliable, this could result in increased costs and delays in the development of our future isotopes, which could adversely affect any future revenue from these future isotopes (assuming receipt of applicable regulatory approvals).
Regulatory approval for production and distribution of radiopharmaceuticals used for medical imaging and therapeutic treatments may involve a lengthy and expensive process with an uncertain outcome.
Currently, the sale or use of Mo-100 is not regulated by a healthcare regulator, such as the FDA, European Medicines Agency (EMA) or comparable foreign regulatory authorities. However, products such as Mo-99 and Tc-99m that are produced from Mo-100 in a cyclotron or a linear accelerator are regulated by healthcare regulators. We expect radiopharmacies, hospitals, clinics and others in the medical community to produce the widely used medical radioisotope technetium-99m (Tc-99m) from the Mo-100 that we may produce using our ASP technology. Tc-99m is a diagnostic agent that is used by health care professionals with FDA-approved imaging devices to detect potential diseases like coronary artery disease and cancer, as well as evaluate lung, liver, kidney and brain function. When used with the appropriate diagnostic scanner device, such as a SPECT imaging system, the Tc-99m emits signals that are captured and produces an image of internal organs to detect various medical problems and contribute to diagnosis and treatment decisions. Our future customers who may use Mo-100 to produce radiopharmaceuticals will likely require regulatory approval for their products. To date, only one healthcare regulator (Canada) has approved the use of Tc-99m produced from Mo-100 via a cyclotron. Obtaining regulatory approval is expensive and can take many years to complete, and its outcome is inherently uncertain. Our customers’ regulatory approval process may not be conducted as planned or completed on schedule, if at all, and failure can occur at any time during the process.
In the future, we may need to obtain approval from the FDA, EMA or comparable foreign regulatory authorities prior to the sale of Mo-100 that we may produce using our ASP technology for use in medical imaging and therapeutic treatments. If we require FDA, EMA or other comparable foreign regulatory authorities to approve the sale of Mo-100 that we may produce using our ASP technology for medical imaging and therapeutic treatments, we must demonstrate the safety and utility or efficacy of our Mo-100. Obtaining regulatory approval is expensive and can take many years to complete, and its outcome is inherently uncertain. Our regulatory approval process may not be conducted as planned or completed on schedule, if at all, and failure can occur at any time during the process.
Other isotopes that we intend to produce in the future may also require approvals from healthcare regulators such as FDA, EMA or comparable foreign regulatory authorities.
| 29 |
| Table of Contents |
Our success depends on our future customers’ ability to successfully commercialize products that are produced from our isotopes.
Our customers operate in a competitive environment. If our customers are unable to successfully commercialize products that they produce from our isotopes, our business will be negatively impacted. Our customers may fail for a number of reasons, including but not limited to pricing pressure from competing products and failure to gain regulatory approval for the production of their products from healthcare regulators.
Our success depends on our ability to adapt to a rapidly changing competitive environment in the nuclear industry.
The nuclear industry in general, and the nuclear fuel industry in particular, is in a period of significant change, which could significantly transform the competitive landscape we face. The uranium and isotope enrichment sector is competitive. Changes in the competitive landscape may adversely affect pricing trends, change customer spending patterns, or create uncertainty. To address these changes, we may seek to adjust our cost structure and efficiency of operations and evaluate opportunities to grow our business organically or through acquisitions and other strategic transactions. We are actively considering, and expect to consider from time to time in the future, potential strategic transactions, which could involve, without limitation, changes in our capital structure, acquisitions and/or dispositions of businesses or assets, joint ventures or investments in businesses, products or technologies.
In connection with any such transaction, we may seek additional debt or equity financing, contribute or dispose of assets, assume additional indebtedness, or partner with other parties to consummate a transaction. Any such transaction may not result in the intended benefits and could involve significant commitments of our financial and other resources. Legal and consulting costs incurred in connection with debt or equity financing transactions in development are deferred and subject to immediate expensing if such a transaction becomes less likely to occur. If the actions we take in response to industry changes are not successful, our business, results of operations and financial condition may be adversely affected.
We may explore strategic collaborations that may never materialize or may fail.
We intend to accelerate the development of our enriched uranium program by selectively collaborating with energy companies in the United States. We intend to retain significant technology, economic and commercial rights to our programs in key geographic areas that are core to our long-term strategy. As a result, we intend to periodically explore a variety of possible additional strategic collaborations in an effort to gain access to additional resources. At the current time, we cannot predict what form such a strategic collaboration might take. We are likely to face significant competition in seeking appropriate strategic collaborators, and negotiations are difficult and time-consuming. We may not be able to negotiate strategic collaborations on acceptable terms, or at all. We are unable to predict when, if ever, we will enter into any additional strategic collaborations because of the numerous risks and uncertainties associated with establishing them.
If the market opportunities for our future enriched isotopes are smaller than we estimate (even assuming receipt of any required regulatory approval), our business may suffer.
We are currently focused on producing enriched isotopes using our ASP technology to meet critical needs in society. We also plan to research the production of enriched uranium using quantum enrichment to meet the future needs of developers of U.S. advanced reactor technologies requiring HALEU. Our projections of the potential markets are based on estimates that have been derived from a variety of sources, including scientific literature and market research, and which may prove to be incorrect. We must be able to successfully acquire a significant market share in our potential markets to achieve profitability and growth. Customers may become difficult to gain access to, which would adversely affect our results of operations and our business.
We face substantial competition, which may result in others discovering, developing or commercializing enriched isotopes before or more successfully than us.
The development and commercialization of radioisotopes and chemical elements is highly competitive. We face competition with respect to all the enriched isotopes that we may produce using our ASP technology from established biotechnology and nuclear medicine technology companies and will face competition with respect to enriched uranium that we may seek to develop or commercialize in the future from innovative technology and energy companies. There are a number of large biotechnology and nuclear medicine technology companies that currently market and sell radioisotopes to radiopharmacies, hospitals, clinics and others in the medical community (Mo-99 is the active ingredient for Tc-99m-based radiopharmaceuticals used in nuclear medicine procedures). There are also a number of technology and energy companies that are currently seeking to develop HALEU. Potential competitors also include academic institutions, government agencies and other public and private research organizations that conduct research, seek patent protection and establish collaborative arrangements for research, development, manufacturing and commercialization.
| 30 |
| Table of Contents |
More established companies may have a competitive advantage over us due to their greater size, resources and institutional experience. In particular, these companies have greater experience and expertise in securing reimbursement, government contracts, relationships with key opinion leaders, obtaining and maintaining regulatory approvals and distribution relationships to market products. These companies also have significantly greater research and marketing capabilities than we do. If we are not able to compete effectively against existing and potential competitors, our business and financial condition may be harmed.
As a result of these factors, our competitors may complete development of isotopes before we are able to, which may limit our ability to develop or commercialize our future isotopes. Our competitors may also develop radioisotopes or technologies that are safer, more effective, more widely accepted and cheaper than ours, and may also be more successful than us in manufacturing and marketing their isotopes. These appreciable advantages could render our future isotopes obsolete or non-competitive before we can recover the expenses of their development and commercialization.
Mergers and acquisitions in the technology and energy industries may result in even more resources being concentrated among a smaller number of our competitors. Smaller and other early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These third parties compete with us in recruiting and retaining qualified scientific, management and commercial personnel, as well as in acquiring technologies complementary to, or necessary for, our programs.
Even if the products that we or our customers may produce using the ASP technology receives regulatory approval, it may fail to achieve market acceptance by radiopharmacies, hospitals, clinics or others in the medical community necessary for commercial success.
Even if the isotopes that we may produce using the ASP technology for the medical industry, or the radioisotopes that we expect our future customers to produce using the stable isotopes that we plan to offer, receives regulatory approval, the isotopes may fail to gain sufficient market acceptance by radiopharmacies, hospitals, clinics and others in the medical community. If it does not achieve an adequate level of acceptance, we may not generate significant product revenue and may not become profitable. The degree of market acceptance of isotopes that we may produce using the ASP technology, or the radioisotopes that our future customers may produce, will depend on a number of factors, including but not limited to:
| · | the potential advantages compared to alternative radioisotopes; |
|
|
|
| · | the timing of market introduction of the product as well as competitive products; |
|
|
|
| · | effectiveness of sales and marketing efforts; |
|
|
|
| · | the strength of our relationships with radiopharmacies, hospitals, clinics and others in the medical community; |
|
|
|
| · | the cost in relation to alternative radioisotopes; |
|
|
|
| · | our ability to offer isotopes that we may produce using the ASP technology for sale at competitive prices; |
|
|
|
| · | the convenience and ease of use compared to alternative radioisotopes; |
|
|
|
| · | the willingness of radiopharmacies, hospitals, clinics and others in the medical community to try an innovative radioisotope; and |
|
|
|
| · | the strength of marketing and distribution support. |
Our efforts to educate radiopharmacies, hospitals, clinics and others in the medical community on the benefits of our isotopes that we may produce using the ASP technology may require significant resources and may never be successful.
Because we expect sales of isotopes that we may produce using the ASP technology (assuming receipt of applicable regulatory approvals for commercial sale) to generate substantially all of our revenues for the foreseeable future, the failure of these isotopes that we may produce using the ASP technology (assuming receipt of applicable regulatory approvals for commercial sale) to find market acceptance would harm our business and could require us to seek additional financing.
| 31 |
| Table of Contents |
We currently have no marketing and sales organization for our future isotopes and have no experience as a company in commercializing products, and we may have to invest significant resources to develop these capabilities. If we are unable to establish marketing and sales capabilities or enter into agreements with third parties to market and sell our products, we may not be able to generate product revenue.
We have no internal sales, marketing or distribution capabilities for our future isotopes, nor have we commercialized any isotopes. If the isotopes that we may produce using our ASP technology gains market acceptance and our customers receive regulatory approval for the isotopes they produce, we must build a marketing and sales organization with technical expertise and supporting distribution capabilities to commercialize such product in the markets that we target, which will be expensive and time-consuming, or collaborate with third parties that have direct sales forces and established distribution systems, either to augment our own sales force and distribution systems or in lieu of our own sales force and distribution systems. We currently plan to independently commercialize the isotopes that we may produce using our ASP technology (assuming receipt of applicable regulatory approvals) in the United States by establishing a focused sales force and marketing infrastructure. We may opportunistically seek additional strategic collaborations to maximize the commercial opportunities for our medical isotopes business outside of the United States. We have no prior experience as a company in the marketing, sale and distribution of isotopes and there are significant risks involved in building and managing a sales organization, including our ability to hire, retain and incentivize qualified individuals, generate sufficient sales leads, provide adequate training to sales and marketing personnel and effectively manage a geographically dispersed sales and marketing team. Any failure or delay in the development of our internal sales, marketing and distribution capabilities would adversely impact the commercialization of these products. We may not be able to enter into collaborations or hire consultants or external service providers to assist us in sales, marketing and distribution functions on acceptable financial terms, or at all. In addition, our product revenues and our profitability, if any, may be lower if we rely on third parties for these functions than if we were to market, sell and distribute any products that we develop ourselves. We likely will have little control over such third parties, and any of them may fail to devote the necessary resources and attention to sell and market our products effectively. If we are not successful in commercializing our isotopes, either on our own or through arrangements with one or more third parties, we may not be able to generate any future product revenue and we would incur significant additional losses.
Obtaining regulatory approval for either the Mo-100 that we may produce using the ASP technology, or the Tc-99m or Mo-99 that our future customers may produce using the Mo-100 that we plan to offer, in one jurisdiction does not mean that we or they will be successful in obtaining regulatory approval of such future products in other jurisdictions.
Currently, the production and distribution of Mo-100 does not require any regulatory licenses from healthcare regulators. Healthcare regulators frequently change such requirements, and it is possible that in the future Mo-100 may be regulated as a healthcare product. Obtaining such regulatory licenses, if required, may be a timely and costly process and could materially impact our ability to commercialize the Mo-100 that we plan to offer. Obtaining regulatory approval of the Mo-100 that we may produce using the ASP technology in one jurisdiction does not guarantee that we will be able to obtain regulatory approval in any other jurisdiction. For example, even if the FDA grants regulatory approval of the Mo-100 that we may produce using the ASP technology, comparable regulatory authorities in foreign jurisdictions must also approve the manufacturing, marketing and promotion and reimbursement of such future product in those countries. However, a failure or delay in obtaining regulatory approval in one jurisdiction may have a negative effect on the regulatory approval process in others. Approval procedures vary among jurisdictions and can involve requirements and administrative review periods different from those in the United States.
Obtaining foreign regulatory approvals and establishing and maintaining compliance with foreign regulatory requirements could result in significant delays, difficulties and costs for us and could delay or prevent the introduction of the Mo-100 that we may produce using the ASP technology. Products such as Tc-99m and Mo-99 that may be produced by our future customers using the Mo-100 that we plan to offer will likely require regulatory licenses in most regions. Healthcare regulators frequently change such requirements and it is unclear what each healthcare regulator will require. To date, only one region (Canada) has approved the use of Tc-99m that has been produced from Mo-100 in a cyclotron. Obtaining such regulatory licenses, if required, may be a timely and costly process and could materially impact the ability of our future customers to operate and use the Mo-100 that we plan to offer. Obtaining regulatory approval in one jurisdiction does not guarantee that we or they will be able to obtain regulatory approval in any other jurisdiction.
If we or any future collaborator fail to comply with the regulatory requirements in international markets or fail to receive applicable marketing approvals, our target market will be reduced and our ability to realize the full market potential of the Mo-100 that we may produce using the ASP technology will be harmed.
Product liability lawsuits against us could cause us to incur substantial liabilities and could limit commercialization of any isotopes that we may produce.
We face an inherent risk of product liability exposure if we commercialize any isotopes that we may produce. If we cannot successfully defend ourselves against claims that any such isotopes caused injuries, we could incur substantial liabilities. Regardless of merit or eventual outcome, liability claims may result in:
| · | decreased demand for any isotopes that we may produce; |
|
|
|
| · | loss of revenue; |
|
|
|
| · | substantial monetary awards to patients; |
|
|
|
| · | significant time and costs to defend the related litigation; |
|
|
|
| · | a diversion of management’s time and our resources; |
|
|
|
| · | initiation of investigations by regulators; |
|
|
|
| · | the inability to commercialize any isotopes that we may produce; |
|
|
|
| · | injury to our reputation and significant negative media attention; and |
|
|
|
| · | a decline in our share price. |
| 32 |
| Table of Contents |
Any product liability insurance coverage that we obtain and maintain may not be adequate to cover all liabilities that we may incur. We anticipate that we will need to increase our insurance coverage each time we commence a clinical trial and if we successfully commercialize any isotopes. Insurance coverage is increasingly expensive. We may not be able to obtain or maintain insurance coverage at a reasonable cost or in an amount adequate to satisfy any liability that may arise.
Risks Related to Regulatory Compliance
Our business is and could become subject to a wide variety of extensive and evolving laws and regulations. Failure to comply with such laws and regulations and failure to obtain licenses, approvals and permits that may be required to execute on our strategy and develop our company’s business could have a material adverse effect on our business.
We are subject to a wide variety of laws and regulations relating to various aspects of our business, including with respect to the development of the ASP technology and our future isotopes, employment and labor, health care, tax, privacy and data security, health and safety, and environmental issues. Laws and regulations at the South African and foreign, federal, state and local levels frequently change, especially in relation to new and emerging industries, and we cannot always reasonably predict the impact of, or the ultimate cost of compliance with, current or future regulatory or administrative changes. In South Africa, our isotope enrichment facilities are heavily regulated. South Africa is a signatory to the International Atomic Energy Agency (“IAEA”) conventions and has adopted safety standards from the IAEA. The design, construction and operation of the isotope enrichment plants are highly regulated and require government licenses, approvals and permits, and may be subject to the imposition of conditions. In some cases, these licenses, approvals and permits entail periodic review and inspections. While we and Klydon have received all licenses, approvals and permits required to build and operate our isotope enrichment facilities in South Africa, we cannot predict whether the conditions associated with such licenses, approvals and permits will be maintained. For example, each of Klydon and ASP Isotopes South Africa (Proprietary) Limited has received from the South African Council for The Non-Proliferation of Weapons of Mass Destruction (1) a registration certificate (which are valid for two years from the date of issuance) and (2) a Manufacturing and Services Permit. The permits provide that the Non-Proliferation Secretariat will conduct at least two industry visits in June and November (or as arranged) of every year. Each of the permits includes numerous conditions, including, for example, the obligation to keep the Council updated or informed on all separation projects at all times and at least through biannual declarations, which must be done through correspondence to the Council at the end of April and September every year. The permit issued to ASP Isotopes South Africa (Proprietary) Limited includes additional specific information requirements related to (i) the progress on the design and construction of the isotope separation plant, (ii) the progress on the manufacturing of isotope separation elements, and (iii) the commissioning of the plant. Each of the permits further provides that (i) any potential export of controlled goods and technology should be requested at an early stage through a Provisional Export Guidance Request, (ii) all isotope separation applications remain controlled regardless of the isotope atomic mass and will be dealt with on a case-by-case basis, and (iii) any ultimate transfer of these controlled goods and technology will be subject to the issuance of a permit by the Council as required in terms of the Non-Proliferation Act and related Government Notices and Regulations.
In addition, we cannot assure you that we will be able to obtain, on a timely basis or at all, any additional licenses, approvals and permits that may be required to execute on our strategy and develop our company’s business, including any such licenses, approvals and permits that may be required to introduce isotopes produced using ASP technology into the market and to begin the enrichment of uranium to demonstrate our capability to produce HALEU using the Quantum Enrichment technology.
Changes in law or the imposition of new or additional regulations or permit requirements that impact our business could negatively impact our performance in various ways, including by limiting our ability to collaborate with partners or customers or by increasing our costs and the time necessary to obtain required authorization. We monitor new developments and devote a significant amount of management’s time and external resources to compliance with these laws and regulations. We cannot assure you, however, that we are and will remain in compliance with all such requirements and, even when we believe we are in compliance, a regulatory agency may determine that we are not. In addition, we cannot assure you that we will be able to obtain all licenses, approvals and permits that may be required to execute on our strategy and develop our company’s business as currently contemplated. Failure by us, our employees, affiliates, partners or others with whom we work to comply with applicable laws and regulations or to obtain or comply with necessary licenses, approvals and permits could result in administrative, civil, commercial or criminal liabilities, including suspension or debarment from government contracts or suspension of our export/import privileges. Failure by us, our employees, affiliates, partners or others with whom we work to comply with the permits issued to us by the South African Council for The Non-Proliferation of Weapons of Mass Destruction could result in disruption of our development activities at our facility in South Africa, which could prevent us from completing our development activities.
| 33 |
| Table of Contents |
If technology developed for the purposes of enriching isotopes can be applied to the creation or development of weapons-grade materials, then our technology may be considered “dual use” technology and be subject to limitations on public disclosure or export.
Our research and development of isotope enrichment is dedicated not only to producing enriched isotopes for use in nuclear medical diagnostic procedures and concentrating uranium in the isotope uranium-235 for use in nuclear energy, but also to safeguarding any information with broad, dual-use potential that could be inappropriately applied. Enrichment is among the most sensitive nuclear technologies because it can produce weapon-grade materials. The ASP technology and the Quantum Enrichment technology may be considered dual use and could be subject to export control, for example, under the Wassenaar Arrangement.
Risks Related to Our Intellectual Property
Our intellectual property is not protected through patents or formal copyright registration. As a result, we do not have the full benefit of patent or copyright laws to prevent others from replicating the ASP technology.
We have not yet protected our intellectual property rights through patents or formal copyright registration, and we currently have no patent applications pending. To date, we have relied exclusively on trade secrets and other intellectual property laws, non-disclosure agreements with our respective employees, consultants, vendors, potential customers and other relevant persons and other measures to protect our intellectual property, and intend to continue to rely on these and other means. As we intend to transition into the commercialization of isotopes, we envision our intellectual property and its security becoming more vital to our future. Until we protect our intellectual property through patent, trademarks and registered copyrights, we may not be able to protect our intellectual property and trade secrets or prevent others from independently developing substantially equivalent proprietary information and techniques or from otherwise gaining access to our intellectual property or trade secrets. In such an instance, our competitors could produce products that are nearly identical to ours, resulting in us selling less products or generating less revenue from our sales.
We may be unable to adequately protect our intellectual property and proprietary rights and prevent others from making unauthorized use of our products and technology.
Our success and competitiveness depend, in significant part, on our ability to protect our intellectual property rights, including the ASP technology and the Quantum Enrichment technology and certain other practices, tools, technologies and technical expertise we utilize in designing, developing, implementing and maintaining processes used in the development of our future isotopes. To date, we have relied exclusively on trade secrets and other intellectual property laws, non-disclosure agreements with our respective employees, consultants, vendors, potential customers and other relevant persons and other measures to protect our intellectual property, and intend to continue to rely on these and other means.
For strategic reasons, we have not yet protected our intellectual property by filing patent applications related to our technology, inventions and improvements. Even if we filed patent applications and patents were granted, we cannot assure you we would be fully protected against third parties as those patents may not be sufficiently broad in their coverage, may not be economically significant, or may not provide us with any competitive advantage. Competitors may be able to design around any patents and develop isotope production techniques comparable or superior to the ASP technology or the Quantum Enrichment technology. Furthermore, the filing of a patent would entail the disclosure of our know-how, and breaches of patent rights related to a wrongful use of this know-how would be difficult to enforce in the international landscape. We believe that our intellectual property strategy differs significantly from the strategies of others involved in the medical isotope industry, many of whom have extensive patent portfolios and rely heavily on intellectual property registrations to enforce their intellectual property rights. As a result of this discrepancy in strategy, we may be at a competitive disadvantage with respect to the strength of our intellectual property protection. Unlike others involved in the medical isotope industry, who generally have patents providing exclusive control over their innovations, we have no recourse against any entity that independently creates the same technology as ours or legitimately reverse-engineers our technology.
We generally enter into non-disclosure agreements with our employees, consultants and other parties with whom we have strategic relationships and business alliances. We cannot, however, assure you that these agreements will be effective in controlling access to and distribution of our technology and proprietary information. Since we do not protect our intellectual property by filing patent applications, we rely on our personnel to protect our trade secrets, know-how and other proprietary information to a greater degree than we would if we had patent protection for our intellectual property. In any jurisdiction in which our research and development is not protected by similar agreements, there is no protection against the manufacture and marketing of identical or comparable research and development by third parties, who are generally free to use, independently develop, and sell our developments and technologies without paying license or royalty fees. Furthermore, our former employees may perform work for our competitors and use our know-how in performing this work. In the event we scale our business by hiring additional personnel and entering into contracts with third parties, the risks associated with breaches of non-disclosure agreements, confidentiality agreements and other agreements pertaining to our technology and proprietary information will increase, and such breaches could have an adverse effect on our business and competitive position.
| 34 |
| Table of Contents |
We may come to believe that third parties are infringing on, or otherwise violating, our intellectual property or other proprietary rights. To prevent infringement or unauthorized use, we may need to file infringement and/or misappropriation suits, which are expensive and time-consuming, could result in meritorious counterclaims against us and would distract management’s attention. In addition, in an infringement or misappropriation proceeding, a court may decide that one or more of our intellectual property rights is invalid, unenforceable, or both, in which case third parties may be able to use our technology without paying license fees or royalties. If we are unable to protect our intellectual property and proprietary rights, we may be unable to prevent competitors from using our own inventions and intellectual property to compete against us, and our business may be harmed.
Our ASP technology may be found to infringe third-party intellectual property rights.
Third parties may in the future assert claims or initiate litigation related to their intellectual property rights in technology that is important to us, including the ASP technology. For example, on October 25, 2022, we received a letter (the “NMS Letter”) from a law firm acting on behalf of Norsk Medisinsk Syklotronsenter AS (“NMS”), asserting, among other things, that the grant of the former license to the ASP technology to us by Klydon violated a pre-existing exclusive sub-license to the ASP technology granted to Radfarma. In November 2023, we entered into a mutual release with NMS, Radfarma, and certain board members and shareholders of Radfarma related to the claims asserted in the NMS letter and other matters, without any payment or license of any rights by any party to the release. Any future claims alleging infringement of intellectual property rights with respect to the ASP technology on which our company relies could be time-consuming, resulting in costly arbitration or litigation and diversion of technical and management personnel, or require us to develop non-infringing technology or enter into license agreements. We cannot assure you that licenses will be available on acceptable terms, if at all. Furthermore, because of the potential for significant damage awards, which are not necessarily predictable, it is not unusual to find even arguably unmeritorious claims resulting in large settlements. If any infringement or other intellectual property claim made against us by any third party is successful, or if we fail to develop non-infringing technology or license the proprietary rights on commercially reasonable terms and conditions, our business, operating results and financial condition could be materially adversely affected.
If the ASP technology infringes the proprietary rights of other parties, we could incur substantial costs, and we may have to take certain actions, including the following:
| · | obtain licenses, which may not be available on commercially reasonable terms, if at all; |
|
|
|
| · | redesign our technology or processes to avoid infringement; |
|
|
|
| · | stop using the subject matter claimed to be held by others; |
|
|
|
| · | pay damages; or |
|
|
|
| · | defend arbitration, litigation or administrative proceedings which may be costly whether we win or lose (and may be prohibitively expensive, particularly for a company of our size), and which could result in a substantial diversion of our financial and management resources. |
In addition, in an infringement proceeding, a court or tribunal may decide that our asserted intellectual property is not valid or is unenforceable. An adverse determination in any litigation, arbitration or defense proceedings could put our intellectual property at risk of being invalidated or interpreted narrowly. If our intellectual property rights are found to be invalid or unenforceable (in whole or in part), our ability to commercialize our future isotopes would suffer and our business, results of operations and financial condition may be adversely affected.
We may enter into collaboration agreements and strategic alliances, and we may not realize the anticipated benefits of such collaborations or alliances.
We may wish to form collaborations in the future with respect to our future isotopes but may not be able to do so or to realize the potential benefits of such transactions, which may cause us to alter or delay our development and commercialization plans. Research and development collaborations are subject to numerous risks, which may include the following:
| · | collaborators have significant discretion in determining the efforts and resources that they will apply to a collaboration and may not commit sufficient efforts and resources or may misapply those efforts and resources; |
|
|
|
| · | collaborators may not pursue development and commercialization of future isotopes or may elect not to continue or renew development or commercialization programs; |
|
|
|
| · | collaborators may delay, provide insufficient resources to, or modify or stop development activities for future isotopes; |
|
|
|
| · | collaborators could develop or acquire products outside of the collaboration that compete directly or indirectly with our future isotopes; |
| 35 |
| Table of Contents |
| · | collaborators may not properly maintain or defend our intellectual property rights or may use our intellectual property or proprietary information in a way that gives rise to actual or threatened litigation that could jeopardize or invalidate our intellectual property or proprietary information or expose us to potential liability; |
|
|
|
| · | disputes may arise between us and a collaborator that cause the delay or termination of the research, development or commercialization of our future isotopes, or that result in costly litigation or arbitration that diverts management attention and resources; |
|
|
|
| · | collaborations may be terminated and, if terminated, may result in a need for additional capital and personnel to pursue further development or commercialization of the applicable future isotopes; and |
|
|
|
| · | collaborators may own or co-own intellectual property covering our products that results from our collaborating with them, and in such cases, we may not have the exclusive right to commercialize such intellectual property. |
The development and potential commercialization of our future isotopes will require substantial additional capital to fund expenses. We may form or seek further strategic alliances, create joint ventures or collaborations, or enter into additional licensing arrangements with third parties that we believe will complement or augment our development and commercialization efforts with respect to our future isotopes, including in territories outside the United States or for certain indications. These transactions can entail numerous operational and financial risks, including exposure to unknown liabilities, disruption of our business and diversion of our management’s time and attention in order to manage a collaboration or develop acquired products or technologies, incurrence of substantial debt or dilutive issuances of equity securities to pay transaction consideration or costs, higher than expected collaboration, acquisition or integration costs, write-downs of assets or goodwill or impairment charges, increased amortization expenses, difficulty and cost in facilitating the collaboration or combining the operations and personnel of any acquired business, impairment of relationships with key suppliers, manufacturers or customers of any acquired business due to changes in management and ownership and the inability to retain key employees of any acquired business. As a result, if we enter into acquisition or in-license agreements or strategic partnerships, we may not be able to realize the benefit of such transactions if we are unable to successfully integrate them with our existing operations and company culture, or if there are materially adverse impacts on our or the counterparty’s operations, which could delay our timelines or otherwise adversely affect our business. We also cannot be certain that, following a strategic transaction or license, we will achieve the revenue or specific net income that justifies such transaction or such other benefits that led us to enter into the arrangement.
In addition, we face significant competition in seeking appropriate strategic partners and the negotiation process is time-consuming and complex. We may not be successful in our efforts to establish a strategic partnership or other alternative arrangements for our future isotopes because they may be deemed to be at too early of a stage of development for collaborative effort and third parties may not view our future isotopes as having the requisite potential to demonstrate safety and efficacy. If and when we collaborate with a third-party for development and commercialization of a future isotope, we can expect to relinquish some or all of the control over the future success of that future isotope to the third-party. Our ability to reach a definitive agreement for a collaboration will depend, among other things, upon our assessment of the collaborator’s resources and expertise, the terms and conditions of the proposed collaboration and the proposed collaborator’s evaluation of our technologies, future isotopes and market opportunities. The collaborator may also consider alternative isotopes or technologies for similar applications that may be available to collaborate on and whether such a collaboration could be more attractive than the one with us for our future isotope. We may also be restricted under any license agreements from entering into agreements on certain terms or at all with potential collaborators.
As a result of these risks, we may not be able to realize the benefit of our existing collaborations or any future collaborations or licensing agreements we may enter into. In addition, we may not be able to negotiate collaborations on a timely basis, on acceptable terms, or at all. If we are unable to do so, we may have to curtail the development of such future isotope, reduce or delay one or more of our other development programs, delay the potential commercialization or reduce the scope of any planned sales or marketing activities for such future isotope, or increase our expenditures and undertake development, manufacturing or commercialization activities at our own expense. If we elect to increase our expenditures to fund development, manufacturing or commercialization activities on our own, we may need to obtain additional capital, which may not be available to us on acceptable terms or at all. If we do not have sufficient funds, we may not be able to further develop our future isotopes or bring them to market and generate product revenue.
| 36 |
| Table of Contents |
We may be dependent on intellectual property licensed or sublicensed to us from, or for which development was funded or otherwise assisted by, government agencies, for development of our technology and future isotopes. Failure to meet our own obligations to any licensor or upstream licensors, including such government agencies, may result in the loss of our rights to such intellectual property, which could harm our business.
Government agencies may provide funding, facilities, personnel or other assistance in connection with the development of the intellectual property rights owned by or licensed to us. Such government agencies may have retained rights in such intellectual property, including the right to grant or require us to grant mandatory licenses or sublicenses to such intellectual property to third parties under certain specified circumstances, including if it is necessary to meet health and safety needs that we are not reasonably satisfying or if it is necessary to meet requirements for public use specified by federal regulations, or to manufacture products in the United States. Any exercise of such rights, including with respect to any such required sublicense of these licenses could result in the loss of significant rights and could harm our ability to commercialize licensed products.
If we are unable to obtain patent protection for our future isotopes, or if the scope of the patent protection obtained is not sufficiently broad, we may not be able to compete effectively in our markets.
We anticipate that we may file patent applications both in the United States and in other countries, as appropriate. However, we cannot predict:
| · | if and when any patents will issue; |
|
|
|
| · | the degree and scope of protection any issued patents will afford us against competitors, including whether third parties will find ways to invalidate or otherwise circumvent our patents; |
|
|
|
| · | whether others will apply for or obtain patents claiming aspects similar to those covered by our patents and patent applications; |
|
|
|
| · | whether we will need to initiate litigation or administrative proceedings to defend our patent rights, which may be costly whether we win or lose; or |
|
|
|
| · | whether the patent applications that we own or in-license will result in issued patents with claims that cover our future isotopes or uses thereof in the United States or in foreign countries. |
We currently rely upon a combination of trade secret protection and confidentiality agreements to protect the intellectual property related to our isotope development techniques and future isotopes. Our success will depend in large part on our ability to obtain and maintain patent protection in the United States and other countries with respect to the ASP technology and the Quantum Enrichment technology. We may seek to protect our proprietary position by filing patent applications in the United States and abroad related to its current and future development programs and future isotopes to the extent permitted by applicable law. The patent prosecution process is expensive and time-consuming, and we may not be able to file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner.
It is possible that we will fail to identify patentable aspects of our research and development output before it is too late to obtain patent protection. The patent applications that we own or in-license may fail to result in issued patents with claims that cover our future isotopes in the United States or in foreign countries. There is no assurance that all of the potentially relevant prior art relating to our patents and patent applications has been found, which can invalidate a patent or prevent a patent from being issued from a pending patent application. Even if patents are successfully issued and even if such patents cover the ASP technology and the Quantum Enrichment technology, third parties may challenge their scope, validity, or enforceability, which may result in such patents being narrowed, invalidated, or held unenforceable. Any successful opposition to these patents or any other patents owned by or licensed to us could deprive us of rights necessary for the successful commercialization of any future isotopes using the ASP technology or the Quantum Enrichment technology. Further, if we encounter delays in regulatory approvals, the period of time during which we could market a future isotope could be reduced.
If the patent applications we hold or have in-licensed with respect to our development programs fail to issue, if their breadth or strength of protection is threatened, or if they fail to provide meaningful exclusivity for the ASP technology or the Quantum Enrichment technology, it could dissuade companies from collaborating with us, and threaten our ability to commercialize, isotopes produced using the ASP technology or the Quantum Enrichment technology. Any such outcome could have a negative effect on our business.
Even if we obtain patents covering the ASP technology or the Quantum Enrichment technology or our methods, we may still be barred from making, using and selling such technology or methods because of the patent rights of others. Others may have filed, and in the future may file, patent applications covering technology or methods that are similar or identical to ours, which could materially affect our ability to successfully develop our technology or to successfully commercialize any isotopes alone or with collaborators.
Patent applications in the United States and elsewhere are generally published approximately 18 months after the earliest filing for which priority is claimed, with such earliest filing date being commonly referred to as the priority date. Therefore, patent applications covering our platform technologies and methods could have been filed by others without our knowledge. Additionally, pending claims in patent applications which have been published can, subject to certain limitations, be later amended in a manner that could cover our platform technologies. These patent applications may have priority over patent applications filed by us.
Obtaining and maintaining our patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by government patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
| 37 |
| Table of Contents |
Periodic maintenance fees, renewal fees, annuity fees and various other government fees on patents and/or applications will be due to be paid to the USPTO and various government patent agencies outside of the United States over the lifetime of our owned and licensed patents and/or applications and any patent rights we may own or license in the future. We will rely on our outside counsel, patent annuity service providers, or our licensing partners to pay these fees due to non-U.S. patent agencies. The USPTO and various non-U.S. government patent agencies require compliance with several procedural, documentary, and other similar provisions during the patent application process. We will employ reputable law firms and other professionals to help us comply. In many cases, an inadvertent lapse can be cured by payment of a late fee or by other means in accordance with the applicable rules. There are situations, however, in which non-compliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. In such an event, potential competitors might be able to enter the market and this circumstance could harm our business.
Third parties may initiate legal proceedings alleging that we are infringing their intellectual property rights, the outcome of which would be uncertain and could have a negative impact on the success of our business.
Our commercial success depends, in part, upon our ability and the ability of our current or future collaborators to develop, manufacture, market and sell our future isotopes and use our proprietary technologies without infringing the proprietary rights and intellectual property of third parties. The technology industry is characterized by extensive and complex litigation regarding patents and other intellectual property rights. Our future isotopes and other proprietary technologies we may develop may infringe existing or future patents owned by third parties. We may in the future become party to, or be threatened with, adversarial proceedings or litigation regarding intellectual property rights with respect to our future isotopes and technology, including interference proceedings, post-grant review and inter partes review before the USPTO. Third parties may assert infringement claims against us based on existing patents or patents that may be granted in the future, regardless of their merit. There is a risk that third parties may choose to engage in litigation with us to enforce or to otherwise assert their patent rights against us. Even if we believe such claims are without merit, a court of competent jurisdiction could hold that these third-party patents are valid, enforceable and infringed, which could have a negative impact on our ability to commercialize our future isotopes. In order to successfully challenge the validity of any such U.S. patent in federal court, we would need to overcome a presumption of validity. As this burden is a high one requiring us to present clear and convincing evidence as to the invalidity of any such U.S. patent claim, there is no assurance that a court of competent jurisdiction would invalidate the claims of any such U.S. patent. If we are found to infringe a third party’s valid and enforceable intellectual property rights, we could be required to obtain a license from such third party to continue developing, manufacturing and marketing our future isotope(s) and technology. However, we may not be able to obtain any required license on commercially reasonable terms or at all. Even if we were able to obtain a license, it could be non-exclusive, thereby giving our competitors and other third parties access to the same technologies licensed to us, and it could require us to make substantial licensing and royalty payments. We could be forced, including by court order, to cease developing, manufacturing and commercializing the infringing technology or future isotope. In addition, we could be found liable for monetary damages, including treble damages and attorneys’ fees, if we are found to have willfully infringed a patent or other intellectual property right. A finding of infringement could prevent us from manufacturing and commercializing our future isotopes or force us to cease some or all of our business operations, which could materially harm our business. Claims that we have misappropriated the confidential information or trade secrets of third parties could have a similar negative impact on our business, financial condition, results of operations and prospects.
Third parties asserting their patent or other intellectual property rights against us may seek and obtain injunctive or other equitable relief, which could effectively block our ability to further develop and commercialize our future isotopes or force us to cease some of our business operations. Defense of these claims, regardless of their merit, would involve substantial litigation expense and would be a substantial diversion of management and other employee resources from our business, cause development delays, and may impact our reputation. In the event of a successful claim of infringement against us, we may have to pay substantial damages, including treble damages and attorneys’ fees for willful infringement, obtain one or more licenses from third parties, pay royalties, or redesign our infringing products, which may be impossible on a cost-effective basis or require substantial time and monetary expenditure. In that event, we would be unable to further develop and commercialize our future isotopes, which could harm our business significantly. Claims that we have misappropriated the confidential information or trade secrets of third parties could have a similar negative impact on our business.
We may be subject to claims asserting that our employees, consultants or advisors have wrongfully used or disclosed alleged trade secrets of their current or former employers or claims asserting ownership of what we regard as our own intellectual property.
Certain of our employees, consultants or advisors are currently, or were previously, employed at universities or other technology companies. Although we try to ensure that our employees, consultants and advisors do not use the proprietary information or know-how of others in their work for us, we may be subject to claims that these individuals or we have used or disclosed intellectual property, including trade secrets or other proprietary information, of any such individual’s current or former employer. Litigation may be necessary to defend against these claims. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. Even if we are successful in defending against such claims, litigation could result in substantial costs and be a distraction to management.
| 38 |
| Table of Contents |
In addition, while it is our policy to require our employees and contractors who may be involved in the conception or development of intellectual property to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who, in fact, conceives or develops intellectual property that we regard as our own. The assignment of intellectual property rights may not be self-executing or the assignment agreements may be breached, and we may be forced to bring claims against third parties, or defend claims that they may bring against us, to determine the ownership of what we regard as our intellectual property.
Reliance on third parties requires us to share our trade secrets, which increases the possibility that a competitor will discover them or that our trade secrets will be misappropriated or disclosed.
If we rely on third parties to manufacture or commercialize our future isotopes, or if we collaborate with additional third parties for the development of our future isotopes, we must, at times, share trade secrets with them. We may also conduct joint research and development programs that may require us to share trade secrets under the terms of our research and development partnerships or similar agreements. We seek to protect our proprietary technology in part by entering into confidentiality agreements and, if applicable, material transfer agreements, services agreements, consulting agreements or other similar agreements with our advisors, employees, third-party contractors and consultants prior to beginning research or disclosing proprietary information. These agreements typically limit the rights of the third parties to use or disclose our confidential information, including our trade secrets. Despite the contractual provisions employed when working with third parties, the need to share trade secrets and other confidential information increases the risk that such trade secrets become known by our competitors, are inadvertently incorporated into the technology of others, or are disclosed or used in violation of these agreements. Given that our proprietary position is based, in part, on our know-how and trade secrets, a competitor’s discovery of our trade secrets or other unauthorized use or disclosure could have an adverse effect on our business and results of operations.
In addition, these agreements typically restrict the ability of our advisors, employees, third-party contractors and consultants to publish data potentially relating to our trade secrets. Despite our efforts to protect our trade secrets, our competitors may discover our trade secrets, either through breach of our agreements with third parties, independent development or publication of information by any third-party collaborators. A competitor’s discovery of our trade secrets could harm our business.
Confidentiality agreements with employees and third parties may not prevent unauthorized disclosure of trade secrets and other proprietary information.
In addition to the protection afforded by patents, we seek to rely on trade secret protection and confidentiality agreements to protect proprietary know-how that is not patentable or that we elect not to patent, processes for which patents are difficult to enforce, and any other elements of our future isotopes, technology and product discovery and development processes that involve proprietary know-how, information, or technology that is not covered by patents. Any disclosure, either intentional or unintentional, by our employees, the employees of third parties with whom we share our facilities or third-party consultants and vendors that we engage, or misappropriation by third parties (such as through a cybersecurity breach) of our trade secrets or proprietary information could enable competitors to duplicate or surpass our technological achievements, thus eroding our competitive position in our market. Because we expect to rely on third parties in the development and manufacture of our future isotopes, we must, at times, share trade secrets with them. Our reliance on third parties requires us to share our trade secrets, which increases the possibility that a competitor will discover them or that our trade secrets will be misappropriated or disclosed.
Trade secrets and confidential information, however, may be difficult to protect. We seek to protect our trade secrets, know-how and confidential information, including our proprietary processes, in part, by entering into confidentiality agreements with our employees, consultants, outside scientific advisors, contractors, and collaborators. With our consultants, contractors, and outside scientific collaborators, these agreements typically include invention assignment obligations. We cannot guarantee that we have entered into such agreements with each party that may have or has had access to our trade secrets or proprietary technology and processes. Although we use reasonable efforts to protect our trade secrets, our employees, consultants, outside scientific advisors, contractors, and collaborators might intentionally or inadvertently disclose our trade secret information to competitors. In addition, competitors may otherwise gain access to our trade secrets or independently develop substantially equivalent information and techniques. Despite these efforts, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, and we may not be able to obtain adequate remedies for such breaches. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. If any of our trade secrets were to be lawfully obtained or independently developed by a competitor or other third-party, we would have no right to prevent them from using that technology or information to compete with us. Furthermore, the laws of some foreign countries do not protect proprietary rights to the same extent or in the same manner as the laws of the United States. As a result, we may encounter significant problems in protecting and defending our intellectual property both in the United States and abroad. If we are unable to prevent unauthorized material disclosure of our intellectual property to third parties, or misappropriation of our intellectual property by third parties, we may not be able to establish or maintain a competitive advantage in our market, which could materially adversely affect our business, operating results, and financial condition.
| 39 |
| Table of Contents |
If we use hazardous and chemical materials in a manner that causes injury or violates applicable law, we may be liable for damages.
Our research and development activities involve the controlled use of potentially hazardous substances, including chemical materials. We are subject to international and local laws and regulations in South Africa governing the use, manufacture, storage, handling and disposal of radioactive and hazardous materials. Although we believe that our procedures for using, handling, storing and disposing of these materials comply with legally prescribed standards, we cannot completely eliminate the risk of contamination or injury resulting from radioactive or hazardous materials. As a result of any such contamination or injury, we may incur liability or local, city, state or federal authorities may curtail the use of these materials and interrupt our business operations. In the event of an accident, we could be held liable for damages or penalized with fines, and the liability could exceed our resources. We do not have any insurance for liabilities arising from radioactive or hazardous materials. Compliance with applicable environmental laws and regulations is expensive, and current or future environmental regulations may impair our research, development, and production efforts, which could harm our business, prospects, financial condition, or results of operations.
Risks Related to Our Business Operations, Employee Matters and Managing Growth
We are highly dependent on the services of our senior management team, and if we are not able to retain these members of our management team and recruit and retain additional management, clinical and scientific personnel, our business will be harmed.
We are highly dependent on our senior management team. The employment agreements we have with these officers do not prevent such persons from terminating their employment with us at any time. The loss of the services of any of these persons could impede the achievement of our research, development and commercialization objectives.
In addition, we will need to attract, retain and motivate highly qualified additional management and scientific personnel. If we are not able to retain our management and to attract, on acceptable terms, additional qualified personnel necessary for the continued development of our business, we may not be able to sustain our operations or grow.
We may not be able to attract or retain qualified personnel in the future due to the intense competition for qualified personnel among biotechnology, pharmaceutical and other businesses. Many of the other pharmaceutical companies that we compete against for qualified personnel and consultants have greater financial and other resources, different risk profiles and a longer history in the industry than we do. They also may provide more diverse opportunities and better chances for career advancement. Some of these characteristics may be more appealing to high-quality candidates and consultants than what we have to offer. If we are unable to continue to attract, retain and motivate high-quality personnel and consultants to accomplish our business objectives, the rate and success at which we can develop future isotopes and our business will be limited, and we may experience constraints on our development objectives.
Our future performance will also depend, in part, on our ability to successfully integrate newly hired executive officers into our management team and our ability to develop an effective working relationship among senior management. Our failure to integrate these individuals and create effective working relationships among them and other members of management could result in inefficiencies in the development and commercialization of our future isotopes, harming future regulatory approvals, sales of our future isotopes and our results of operations. Additionally, we do not currently maintain “key person” life insurance on the lives of our executives or any of our employees.
We will need to expand our organization, and we may experience difficulties in managing this growth, which could disrupt our operations.
As of December 31, 2023, we employed 76 people on a full-time basis, 69 of whom are located in South Africa. We rely on service providers for certain general administrative, financial, accounting, tax, intellectual property and other legal services, and we will need to expand our organization to hire qualified personnel to perform these functions internally. Our management may need to divert significant attention and time to managing these growth activities. We may not be able to effectively manage the expansion of our operations, which may result in weaknesses in our infrastructure, operational inefficiencies, loss of business opportunities, loss of employees and reduced productivity among remaining employees. Our expected growth could require significant capital expenditures and may divert financial resources from other projects, such as the development of our future isotopes. If our management is unable to effectively manage our growth, our expenses may increase more than expected, our ability to generate and grow revenue could be reduced and we may not be able to implement our business strategy. Our future financial performance, our ability to commercialize future isotopes, develop a scalable infrastructure and compete effectively will depend, in part, on our ability to effectively manage any future growth.
| 40 |
| Table of Contents |
Our employees, consultants and commercial partners may engage in misconduct or other improper activities, including non-compliance with regulatory standards and requirements and insider trading.
We are exposed to the risk of fraud or other misconduct by our employees, consultants and commercial partners. Misconduct by these parties could include intentional failures to comply with FDA regulations or the regulations applicable in other jurisdictions, provide accurate information to the FDA and other regulatory authorities, report financial information or data accurately or disclose unauthorized activities to us. It is not always possible to identify and deter employee misconduct, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from government investigations or other actions or lawsuits stemming from a failure to comply with these laws or regulations. If any such actions are instituted against us and we are not successful in defending ourselves or asserting our rights, those actions could have a negative impact on our business, financial condition, results of operations and prospects, including the imposition of significant fines or other sanctions.
We and our contractors are highly dependent on the performance of sub-contractors and other third parties.
We and our contractors are highly dependent on the performance of sub-contractors and other third parties. If these contractors, sub-contractors and third parties are unable to deliver the results that we require, our operating results could be adversely affected and our business could be materially harmed.
Significant disruptions of our information technology systems or data security incidents could result in significant financial, legal, regulatory, business and reputational harm to us.
We are dependent on information technology systems and infrastructure, including mobile technologies, to operate our business. In the ordinary course of our business, we collect, store, process and transmit large amounts of sensitive information, including intellectual property, proprietary business information, personal information and other confidential information. It is critical that we do so in a secure manner to maintain the confidentiality, integrity and availability of such sensitive information. We have also outsourced elements of our operations (including elements of our information technology infrastructure) to third parties, and as a result, we manage a number of third-party vendors who may or could have access to our computer networks or our confidential information. In addition, many of those third parties, in turn, subcontract or outsource some of their responsibilities to third parties. While all information technology operations are inherently vulnerable to inadvertent or intentional security breaches, incidents, attacks and exposures, the accessibility and distributed nature of our information technology systems, and the sensitive information stored on those systems, make such systems potentially vulnerable to unintentional or malicious, internal and external attacks on our technology environment. In addition, some of our employees work remotely, which may make us more vulnerable to cyberattacks. Potential vulnerabilities can be exploited from inadvertent or intentional actions of our employees, third-party vendors, business partners, or by malicious third parties. Attacks of this nature are increasing in their frequency, levels of persistence, sophistication and intensity, and are being conducted by sophisticated and organized groups and individuals with a wide range of motives (including, but not limited to, industrial espionage) and expertise, including organized criminal groups, “hacktivists,” nation states and others. In addition to the extraction of sensitive information, such attacks could include the deployment of harmful malware, ransomware, denial-of-service attacks, social engineering and other means to affect service reliability and threaten the confidentiality, integrity and availability of information. In addition, the prevalent use of mobile devices increases the risk of data security incidents.
Significant disruptions of our, our third-party vendors’ and/or our business partners’ information technology systems or other similar data security incidents could adversely affect our business operations and/or result in the loss, misappropriation, and/or unauthorized access, use or disclosure of, or the prevention of access to, sensitive information, which could result in financial, legal, regulatory, business and reputational harm to us. In addition, information technology system disruptions, whether from attacks on our technology environment or from computer viruses, natural disasters, terrorism, war and telecommunication and electrical failures, could result in a material disruption of our development programs and our business operations. Additionally, theft of our intellectual property or proprietary business information could require substantial expenditures to remedy. If we or our third-party collaborators, consultants, contractors, suppliers, or service providers were to suffer an attack or breach, for example, that resulted in the unauthorized access to or use or disclosure of personal or health information, we may have to notify consumers, partners, collaborators, government authorities, and the media, and may be subject to investigations, civil penalties, administrative and enforcement actions, and litigation, any of which could harm our business and reputation.
| 41 |
| Table of Contents |
There is no way of knowing with certainty whether we have experienced any data security incidents that have not been discovered. While we have no reason to believe this to be the case, attackers have become very sophisticated in the way they conceal access to systems, and many companies that have been attacked are not aware that they have been attacked. Any event that leads to unauthorized access, use or disclosure of personal information, including but not limited to personal information regarding our patients or employees, could disrupt our business, harm our reputation, compel us to comply with applicable federal and/or state breach notification laws and foreign law equivalents, subject us to time-consuming, distracting and expensive litigation, regulatory investigation and oversight, mandatory corrective action, require us to verify the correctness of database contents, or otherwise subject us to liability under laws, regulations and contractual obligations, including those that protect the privacy and security of personal information. This could result in increased costs to us, and result in significant legal and financial exposure and/or reputational harm. In addition, any failure or perceived failure by us or our vendors or business partners to comply with our privacy, confidentiality or data security-related legal or other obligations to third parties, or any further security incidents or other inappropriate access events that result in the unauthorized access, release or transfer of sensitive information, which could include personally identifiable information, may result in governmental investigations, enforcement actions, regulatory fines, litigation, or public statements against us by advocacy groups or others, and could cause third parties, including clinical sites, regulators or current and potential partners, to lose trust in us or we could be subject to claims by third parties that we have breached our privacy- or confidentiality-related obligations, which could materially and adversely affect our business and prospects. Moreover, data security incidents and other inappropriate access can be difficult to detect, and any delay in identifying them may lead to increased harm of the type described above. While we have implemented security measures intended to protect our information technology systems and infrastructure, there can be no assurance that such measures will successfully prevent service interruptions or security incidents.
Our international operations subject us to risks of doing business in foreign countries, which could adversely affect our business, financial condition and results of operations.
Our primary operations are located outside the U.S. (primarily the construction of isotope enrichment plants in South Africa), and we plan to sell our isotopes to customers outside the U.S. Accordingly, our business is subject to risks related to the differing legal, political, social and regulatory requirements and economic conditions of non-U.S. jurisdictions. Risks inherent in international operations include the following:
| · | fluctuations in foreign currency exchange rates may affect product demand and may adversely affect the profitability in U.S. dollars of products and services we provide in international markets where payment for our products and services is made in the local currency; |
|
|
|
| · | transportation and other shipping costs may increase, or transportation may be inhibited; |
|
|
|
| · | increased cost or decreased availability of raw materials; |
|
|
|
| · | changes in foreign laws and tax rates or U.S. laws and tax rates with respect to foreign income may unexpectedly increase the rate at which our income is taxed, impose new and additional taxes on remittances, repatriation or other payments by subsidiaries, or cause the loss of previously recorded tax benefits; |
|
|
|
| · | foreign countries in which we do business may adopt other restrictions on foreign trade or investment, including currency exchange controls; |
|
|
|
| · | trade sanctions by or against these countries could result in our losing access to customers and suppliers in those countries; |
|
|
|
| · | unexpected adverse changes in foreign laws or regulatory requirements may occur; |
|
|
|
| · | our agreements with counterparties in foreign countries may be difficult for us to enforce and related receivables may be difficult for us to collect; |
|
|
|
| · | compliance with the variety of foreign laws and regulations may be unduly burdensome; |
|
|
|
| · | compliance with anti-bribery and anti-corruption laws (such as the Foreign Corrupt Practices Act) as well as anti-money- laundering laws may be costly; |
|
|
|
| · | unexpected adverse changes in export duties, quotas and tariffs and difficulties in obtaining export licenses may occur; |
|
|
|
| · | general economic conditions in the countries in which we operate could have an adverse effect on our earnings from operations in those countries; |
|
|
|
| · | our foreign operations may experience staffing difficulties and labor disputes; |
|
|
|
| · | termination or substantial modification of international trade agreements may adversely affect our access to raw materials and to markets for our products outside the U.S.; |
|
|
|
| · | foreign governments may nationalize or expropriate private enterprises; |
|
|
|
| · | increased sovereign risk (such as default by or deterioration in the economies and creditworthiness of local governments) may occur; and |
|
|
|
| · | political or economic repercussions from terrorist activities, including the possibility of hyperinflationary conditions and political instability, may occur in certain countries in which we do business. |
Unanticipated events, such as geopolitical changes, could result in a write-down of our investment in the affected joint venture or a delay or cause cancellation of those capital projects, which could negatively impact our future growth and profitability. Our success as a global business will depend, in part, upon our ability to succeed in differing legal, regulatory, economic, social and political conditions by developing, implementing and maintaining policies and strategies that are effective in each location where we and our joint ventures do business.
| 42 |
| Table of Contents |
Furthermore, we will be subject to rules and regulations related to anti-bribery and anti-trust prohibitions of the U.S. and other countries, as well as export controls and economic embargoes, violations of which may carry substantial penalties. For example, export control and economic embargo regulations limit the ability of our subsidiaries to market, sell, distribute or otherwise transfer their products or technology to prohibited countries or persons. Failure to comply with these regulations could subject our subsidiaries to fines, enforcement actions and/or have an adverse effect on our reputation and the value of our Common Stock.
Our tangible assets may be subject to defects in title.
We have investigated our rights to the assets we have purchased and developed, and, to the best of our knowledge, those rights are in good standing. However, no assurance can be given that such rights will not be revoked, or significantly altered, to our detriment. There can also be no assurance that our rights will not be challenged or impugned by third parties, including by governments and non-governmental organizations.
We are subject to foreign currency risks.
Our operations are subject to foreign currency fluctuations. Our current operating expenses are primarily transacted in U.S. dollars, while our current revenues and some of our cash balances and expenses are measured in other currencies. As our business expands internationally, the U.S. dollar may or may not be our primary current for operating expenses. Any strengthening or weakening of the U.S. dollar in relation to the currencies of other countries or vice versa can have a material impact on our cash flows and profitability and affect the value of our assets and shareholders’ equity.
Risks Related to Ownership of Our Common Stock
We do not know whether an active, liquid and orderly trading market will develop for our Common Stock or what the market price of our Common Stock will be and as a result it may be difficult for you to sell your shares of our Common Stock.
Prior to our IPO in November of 2022, there was no public market for shares of our Common Stock. Although our Common Stock is listed on the Nasdaq Capital Market (Nasdaq), only a limited trading market for our shares has developed, and an active market may never develop or if developed be sustained in the future. You may not be able to sell your shares quickly or at the market price if trading in shares of our Common Stock is not active. Further, an inactive market may also impair our ability to raise capital by selling shares of our Common Stock and may impair our ability to enter into strategic partnerships or acquire companies or products by using our shares of Common Stock as consideration.
The price of our stock may be volatile, and you could lose all or part of your investment.
The trading price of our Common Stock is volatile and could be subject to wide fluctuations in response to various factors, some of which are beyond our control, including limited trading volume. In addition to the factors discussed in this “Risk Factors” section and elsewhere in this Annual Report on Form 10-K, these factors include:
| · | adverse results or delays in our development activities; |
|
|
|
| · | adverse regulatory decisions, including failure to receive regulatory approval for our future isotopes; |
|
|
|
| · | changes in laws or regulations applicable to our future isotopes, including but not limited to requirements for approvals; |
|
|
|
| · | any changes to our relationship with any manufacturers, suppliers, licensors, future collaborators or other strategic partners; |
|
|
|
| · | our inability to obtain adequate product supply for any future isotope or inability to do so at acceptable prices; |
|
|
|
| · | our inability to establish collaborations if needed; |
|
|
|
| · | our failure to commercialize our future isotopes; |
|
|
|
| · | additions or departures of key scientific or management personnel; |
|
|
|
| · | unanticipated serious safety concerns related to the use of our future isotopes; |
|
|
|
| · | introduction of new products or services offered by us or our competitors; |
|
|
|
| · | announcements of significant acquisitions, strategic partnerships, joint ventures, or capital commitments by us or our competitors; |
|
|
|
| · | our ability to effectively manage our growth; |
|
|
|
| · | actual or anticipated variations in quarterly operating results; |
|
|
|
| · | our cash position; |
|
|
|
| · | our failure to meet the estimates and projections of the investment community or that we may otherwise provide to the public; |
| 43 |
| Table of Contents |
| · | publication of research reports about us or our industry or positive or negative recommendations or withdrawal of research coverage by securities analysts; |
|
|
|
| · | changes in the market valuations of similar companies; |
|
|
|
| · | overall performance of the equity markets; |
|
|
|
| · | issuances of debt or equity securities; |
|
|
|
| · | sales of our Common Stock by us or our stockholders in the future or the perception that such sales may occur; |
|
|
|
| · | trading volume of our Common Stock; |
|
|
|
| · | changes in accounting practices; |
|
|
|
| · | ineffectiveness of our internal controls; |
|
|
|
| · | disputes or other developments relating to proprietary rights, including patents, litigation matters, and our ability to obtain patent protection for our technologies; |
|
|
|
| · | significant lawsuits, including patent or stockholder litigation; |
|
|
|
| · | general political and economic conditions, including military conflict or the COVID-19 pandemic; and |
|
|
|
| · | other events or factors, many of which are beyond our control. |
In addition, the stock market in general, and biopharmaceutical companies in particular, have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies. Broad market and industry factors may negatively affect the market price of our Common Stock, regardless of our actual operating performance, and you may not realize any return on your investment in us and may lose some or all of your investment. In the past, securities class action litigation has often been instituted against companies following periods of volatility in the market price of a company’s securities. This type of litigation, if instituted, could result in substantial costs and a diversion of management’s attention and resources, which would harm our business, operating results or financial condition.
We do not intend to pay dividends on our Common Stock, so any returns will be limited to the value of our stock.
We have never declared or paid any cash dividend on our Common Stock. We currently anticipate that we will retain future earnings for the development, operation, and expansion of our business and do not anticipate declaring or paying any cash dividends for the foreseeable future. Any return to stockholders would therefore be limited to the appreciation, if any, of their stock.
Our principal stockholders and management own a significant percentage of our stock and will be able to exert significant control over matters subject to stockholder approval.
Our executive officers, current directors, greater than 5% holders, and their affiliates beneficially own, in the aggregate, approximately 43.3% of our Common Stock as of December 31, 2023. Therefore, these stockholders will have the ability to influence us through this ownership position. These stockholders may be able to determine all matters requiring stockholder approval. For example, these stockholders may be able to control elections of directors, amendments of our organizational documents, or approval of any merger, sale of assets, or other major corporate transaction. This may prevent or discourage unsolicited acquisition proposals or offers for our Common Stock that you may feel are in your best interest as one of our stockholders.
Sales of a substantial number of shares of our Common Stock by our existing stockholders in the public market, or the perception that such sales could occur, could cause our stock price to fall.
As of April 1, 2024, we had a total of 48,923,276 shares of Common Stock outstanding. If our existing stockholders sell, or indicate an intention to sell, substantial amounts of our Common Stock in the public market, the trading price of our Common Stock could decline.
Of our outstanding Common Stock, the shares held by directors, executive officers, and other affiliates are subject to volume limitations under Rule 144 under the Securities Act. In addition, 3,254,606 shares of Common Stock that are either subject to outstanding options or reserved for future issuance under our employee benefit plans will become eligible for sale in the public market to the extent permitted by the provisions of various vesting schedules, and Rule 144 and Rule 701 under the Securities Act. If these additional shares of Common Stock are sold, or if it is perceived that they will be sold in the public market, the trading price of our Common Stock could decline. Any sales of securities by our stockholders could have a material adverse effect on the trading price of our Common Stock.
| 44 |
| Table of Contents |
Future sales and issuances of our Common Stock or rights to purchase Common Stock, including pursuant to our equity incentive plans, could result in additional dilution of the percentage ownership of our stockholders and could cause our stock price to fall.
We expect that we will need significant additional capital in the future to continue our planned operations, including development activities, commercialization efforts if we are able to obtain marketing approval of future isotopes, research and development activities, and costs associated with operating a public company. To raise capital, we may sell Common Stock, convertible securities or other equity securities in one or more transactions at prices and in a manner that we determine from time to time. If we sell Common Stock, convertible securities or other equity securities, investors may be materially diluted by subsequent sales. Such sales may also result in material dilution to our existing stockholders, and new investors could gain rights, preferences and privileges senior to the holders of our Common Stock.
Pursuant to our 2022 Plan, our management is authorized to grant stock options to our employees, directors and consultants. Additionally, the number of shares of our Common Stock reserved for issuance under our 2022 Plan will automatically increase on January 1 of each year, beginning on January 1, 2023 and continuing through and including January 1, 2032, by 5% of the total number of shares of our capital stock outstanding on December 31 of the preceding calendar year (determined on an as-converted to voting Common Stock basis, without regard to any limitations on the conversion of the non-voting Common Stock), or a lesser number of shares determined by our board of directors. Such issuances will result in dilution to our stockholders.
We have broad discretion in the use of our existing cash and cash equivalents and may not use them effectively.
Our management has broad discretion in the application of our existing cash and cash equivalents. Because of the number and variability of factors that will determine our use of our existing cash and cash equivalents, their ultimate use may vary substantially from their currently intended use. Our management might not apply our existing cash and cash equivalents in ways that ultimately increase the value of our Common Stock. The failure by our management to apply these funds effectively could harm our business. We intend to invest our existing cash and cash equivalents that are not used as described above in short- and medium-term, investment-grade, interest-bearing instruments, certificates of deposit or direct or guaranteed obligations of the U.S. government. These investments may not yield a favorable return to our stockholders. If we do not invest or apply our existing cash and cash equivalents in ways that enhance stockholder value, we may fail to achieve expected financial results, which could cause our stock price to decline.
We are an emerging growth company and a smaller reporting company, and the reduced reporting requirements applicable to emerging growth companies and smaller reporting companies may make our Common Stock less attractive to investors.
We are an emerging growth company, as defined in the JOBS Act. For as long as we continue to be an emerging growth company, we may take advantage of exemptions from various reporting requirements that are applicable to other public companies that are not emerging growth companies, including not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements and exemptions from the requirements of holding nonbinding advisory votes on executive compensation and stockholder approval of any golden parachute payments not previously approved. We could be an emerging growth company until December 31, 2027, although circumstances could cause us to lose that status earlier, including if we become a “large accelerated filer” as defined in Rule 12b-2 under the Exchange Act or if we have total annual gross revenue of $1.235 billion or more during any fiscal year before that time, in which cases we would no longer be an emerging growth company as of the following December 31 or, if we issue more than $1.0 billion in non-convertible debt during any three year period before that time, we would cease to be an emerging growth company immediately. Even after we no longer qualify as an emerging growth company, we may still qualify as a “smaller reporting company” which would allow us to take advantage of many of the same exemptions from disclosure requirements including not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act and reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements. Investors may find our Common Stock less attractive because we may rely on these exemptions. If some investors find our Common Stock less attractive as a result, there may be a less active trading market for our Common Stock and our stock price may be more volatile.
In addition, the JOBS Act provides that an emerging growth company can take advantage of an extended transition period for complying with new or revised accounting standards. This provision allows an emerging growth company to delay the adoption of accounting standards that have different effective dates for public and private companies until those standards would otherwise apply to private companies. We have elected to avail ourselves of this exemption from new or revised accounting standards, and therefore we will not be subject to the same requirements to adopt new or revised accounting standards as other public companies that are not emerging growth companies.
We are also a “smaller reporting company” as defined in the Exchange Act. We may continue to be a smaller reporting company even after we are no longer an emerging growth company. We may take advantage of certain of the scaled disclosures available to smaller reporting companies and will be able to take advantage of these scaled disclosures for so long as our voting and non-voting common stock held by non-affiliates is less than $250.0 million measured on the last business day of our second fiscal quarter, or our annual revenue is less than $100.0 million during the most recently completed fiscal year and our voting and non-voting common stock held by non-affiliates is less than $700.0 million measured on the last business day of our second fiscal quarter.
| 45 |
| Table of Contents |
Delaware law and provisions in our certificate of incorporation and bylaws, as amended, could make a merger, tender offer or proxy contest difficult, thereby depressing the trading price of our common stock.
Provisions of certificate of incorporation and bylaws as amended may delay or discourage transactions involving an actual or potential change in our control or change in our management, including transactions in which stockholders might otherwise receive a premium for their shares or transactions that our stockholders might otherwise deem to be in their best interests. Therefore, these provisions could adversely affect the price of our common stock. Among other things, our amended and restated certificate of incorporation and amended and restated bylaws:
| · | permit our board of directors to issue up to 10,000,000 shares of preferred stock, with any rights, preferences and privileges as they may designate (including the right to approve an acquisition or other change in our control); |
|
|
|
| · | provide that the authorized number of directors may be changed only by resolution of the board of directors; |
|
|
|
| · | provide that our board of directors or any individual director may only be removed with cause and the affirmative vote of the holders of at least 66-2/3% of the voting power of all of our then-outstanding shares of the capital stock entitled to vote generally in the election of directors, voting together as a single class; |
|
|
|
| · | provide that all vacancies, including newly created directorships, may, except as otherwise required by law, be filled by the affirmative vote of a majority of directors then in office, even if less than a quorum; |
|
|
|
| · | divide our board of directors into three classes; |
|
|
|
| · | require that any action to be taken by our stockholders must be effected at a duly called annual or special meeting of stockholders and not be taken by written consent; |
|
|
|
| · | provide that stockholders seeking to present proposals before a meeting of stockholders or to nominate candidates for election as directors at a meeting of stockholders must provide notice in writing in a timely manner and also specify requirements as to the form and content of a stockholder’s notice; |
|
|
|
| · | do not provide for cumulative voting rights (therefore allowing the holders of a majority of the shares of common stock entitled to vote in any election of directors to elect all of the directors standing for election, if they should so choose); |
|
|
|
| · | provide that special meetings of our stockholders may be called only by the chair of our board of directors, our Chief Executive Officer or by the board of directors pursuant to a resolution adopted by a majority of the total number of authorized directors; and |
|
|
|
| · | provide that the Court of Chancery of the State of Delaware will be the sole and exclusive forum for the following types of actions or proceedings under state, statutory and common law: (i) any derivative action or proceeding brought on our behalf; (ii) any action asserting a claim of breach of a fiduciary duty owed by any of our directors, officers or other employees to us or our stockholders; (iii) any action asserting a claim pursuant to any provision of the Delaware General Corporation Law, our certificate of incorporation or our bylaws; (iv) any action as to which the Delaware General Corporation Law confers jurisdiction to the Court of Chancery of the State of Delaware; and (v) any action governed by the internal affairs doctrine, in all cases subject to the court’s having personal jurisdiction over the indispensable parties named as defendants; provided these provisions of our amended and restated certificate of incorporation and amended and restated bylaws will not apply to suits brought to enforce a duty or liability created by the Exchange Act, or any other claim for which the federal courts have exclusive jurisdiction; and provided that, unless we consent in writing to the selection of an alternative forum, to the fullest extent permitted by law, the federal district courts of the United States of America shall be the exclusive forum for the resolution of any complaint asserting a cause of action arising under the Securities Act of 1933, as amended (Securities Act), including all causes of action asserted against any defendant to such complaint. For the avoidance of doubt, this provision is intended to benefit and may be enforced by us, our officers and directors, the underwriters to any offering giving rise to such complaint, and any other professional or entity whose profession gives authority to a statement made by that person or entity and who has prepared or certified any part of the documents underlying the offering. |
These provisions, with the exception of the ability of our board of directors to issue shares of preferred stock and designate any rights, preferences and privileges thereto, would require approval by the holders of at least 66-2/3% of our then-outstanding common stock.
In addition, as a Delaware corporation, we are subject to Section 203 of the Delaware General Corporation Law. These provisions may prohibit large stockholders, in particular those owning 15% or more of our outstanding voting stock, from merging or combining with us for a certain period of time. A Delaware corporation may opt out of this provision by express provision in its original certificate of incorporation or by amendment to its certificate of incorporation or bylaws approved by its stockholders. However, we have not opted out of this provision.
| 46 |
| Table of Contents |
These and other provisions in our certificate of incorporation and bylaws, as amended, and Delaware law could make it more difficult for stockholders or potential acquirors to obtain control of our board of directors or initiate actions that are opposed by our then-current board of directors, including delay or impede a merger, tender offer or proxy contest involving our company. The existence of these provisions could negatively affect the price of our common stock and limit opportunities for you to realize value in a corporate transaction.
Our amended and restated certificate of incorporation provides that the Court of Chancery of the State of Delaware will be the exclusive forum for certain disputes between us and our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or our directors, officers, or employees.
Our amended and restated certificate of incorporation provides that, subject to the court’s having personal jurisdiction over the indispensable parties named as defendants, unless we consent in writing to the selection of an alternative forum, the Court of Chancery of the State of Delaware will be the sole and exclusive forum for the following types of actions or proceedings:
| · | any derivative action or proceeding brought on our behalf; |
|
|
|
| · | any action asserting a claim of breach of fiduciary duty owed by any of our directors, officers or other employees to us or our stockholders; |
|
|
|
| · | any action asserting a claim arising pursuant to any provision of the Delaware General Corporation Law, our certificate of incorporation or bylaws; |
|
|
|
| · | any action as to which the Delaware General Corporation Law confers jurisdiction to the Court of Chancery of the State of Delaware; and |
|
|
|
| · | any action asserting a claim that is governed by the internal affairs doctrine. |
This provision would not apply to suits brought to enforce a duty or liability created by the Exchange Act. Furthermore, Section 22 of the Securities Act creates concurrent jurisdiction for federal and state courts over all such Securities Act actions. Accordingly, both state and federal courts have jurisdiction to entertain such claims. To prevent having to litigate claims in multiple jurisdictions and the threat of inconsistent or contrary rulings by different courts, among other considerations, our amended and restated certificate of incorporation further provides that the federal district courts of the United States of America will be the exclusive forum for resolving any complaint asserting a cause of action arising under the Securities Act. While the Delaware courts have determined that such choice of forum provisions are facially valid, a stockholder may nevertheless seek to bring a claim in a venue other than those designated in the exclusive forum provisions. In such instance, we would expect to vigorously assert the validity and enforceability of the exclusive forum provisions of our amended and restated certificate of incorporation. This may require significant additional costs associated with resolving such action in other jurisdictions and there can be no assurance that the provisions will be enforced by a court in those other jurisdictions.
These exclusive forum provisions may limit a stockholder’s ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors, officers, or other employees, which may discourage lawsuits against us and our directors, officers and other employees. If a court were to find either exclusive forum provision in our amended and restated certificate of incorporation to be inapplicable or unenforceable in an action, we may incur further significant additional costs associated with resolving the dispute in other jurisdictions, all of which could seriously harm our business.
We are currently listed on The Nasdaq Capital Market. If we are unable to maintain listing of our securities on Nasdaq or any stock exchange, our stock price could be adversely affected and the liquidity of our stock and our ability to obtain financing could be impaired and it may be more difficult for our shareholders to sell their securities.
Although our Common Stock is currently listed on The Nasdaq Capital Market, we may not be able to continue to meet the exchange’s minimum listing requirements or those of any other national exchange. If we are unable to maintain listing on Nasdaq or if a liquid market for our Common Stock does not develop or is sustained, our Common Stock may remain thinly traded.
The Listing Rules of Nasdaq require listing issuers to comply with certain standards in order to remain listed on its exchange. If, for any reason, we should fail to maintain compliance with these listing standards and Nasdaq should delist our securities from trading on its exchange and we are unable to obtain listing on another national securities exchange, a reduction in some or all of the following may occur, each of which could have a material adverse effect on our shareholders:
| · | the liquidity of our Common Stock; |
|
|
|
| · | the market price of our Common Stock; |
|
|
|
| · | our ability to obtain financing for the continuation of our operations; |
|
|
|
| · | the number of investors that will consider investing in our Common Stock; |
|
|
|
| · | the number of market makers in our Common Stock; |
|
|
|
| · | the availability of information concerning the trading prices and volume of our Common Stock; and |
|
|
|
| · | the number of broker-dealers willing to execute trades in shares of our Common Stock. |
| 47 |
| Table of Contents |
General Risk Factors
We will incur significant increased costs as a result of operating as a public company, and our management will be required to devote substantial time to new compliance initiatives.
We became a public company in November of 2022, and as a public company we will incur significant legal, accounting, and other expenses that we did not incur as a private company. We are subject to the reporting requirements of the Exchange Act, which require, among other things, that we file with the SEC annual, quarterly, and current reports with respect to our business and financial condition. In addition, the Sarbanes-Oxley Act, as well as rules subsequently adopted by the SEC and Nasdaq to implement provisions of the Sarbanes-Oxley Act, impose significant requirements on public companies, including requiring establishment and maintenance of effective disclosure and financial controls and changes in corporate governance practices. Further, in July 2010, the Dodd-Frank Wall Street Reform and Consumer Protection Act (Dodd-Frank Act) was enacted. There are significant corporate governance and executive compensation-related provisions in the Dodd-Frank Act that require the SEC to adopt additional rules and regulations in these areas such as “say on pay” and proxy access. Emerging growth companies and smaller reporting companies are exempted from certain of these requirements, but we may be required to implement these requirements sooner than budgeted or planned and thereby incur unexpected expenses. Stockholder activism, the current political environment and the current high level of government intervention and regulatory reform may lead to substantial new regulations and disclosure obligations, which may lead to additional compliance costs and impact the manner in which we operate our business in ways we cannot currently anticipate.
We expect the rules and regulations applicable to public companies to substantially increase our legal and financial compliance costs and to make some activities more time-consuming and costly. If these requirements divert the attention of our management and personnel from other business concerns, they could have a material adverse effect on our business, financial condition, and results of operations. The increased costs will decrease our net income or increase our net loss, and may require us to reduce costs in other areas of our business or increase the prices of our products or services. For example, we expect these rules and regulations to make it more difficult and more expensive for us to obtain director and officer liability insurance and we may be required to incur substantial costs to maintain the same or similar coverage. We cannot predict or estimate the amount or timing of additional costs we may incur to respond to these requirements. The impact of these requirements could also make it more difficult for us to attract and retain qualified persons to serve on our board of directors, our board committees or as executive officers.
We have identified a material weakness in our internal control over financial reporting. If our remediation of this material weakness is not effective, or if we experience material weaknesses in the future or otherwise fail to implement and maintain an effective system of internal controls in the future, we may not be able to accurately report our financial condition or results of operations which may adversely affect investor confidence in us, and as a result, the value of our Common Stock.
Our Common Stock was listed on the Nasdaq Stock Exchange on November 10, 2022. Prior to listing, we were a privately-held company, we were not required to evaluate our internal control over financial reporting in a manner that meets the standards of publicly traded companies required by Section 404(a) of the Sarbanes-Oxley Act, or Section 404. As a public company, we are subject to significant requirements for enhanced financial reporting and internal controls. The process of designing and implementing effective internal controls is a continuous effort that requires us to anticipate and react to changes in our business and the economic and regulatory environments and to expend significant resources to maintain a system of internal controls that is adequate to satisfy our reporting obligations as a public company. In addition, we are required, pursuant to Section 404, to furnish a report by management on, among other things, the effectiveness of our internal control over financial reporting in the annual report. This assessment includes disclosure of any material weaknesses identified by our management in our internal control over financial reporting. A material weakness is a deficiency or combination of deficiencies in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of a company’s annual and interim financial statements will not be detected or prevented on a timely basis.
The rules governing the standards that must be met for our management to assess our internal control over financial reporting are complex and require significant documentation, testing, and possible remediation. Testing and maintaining internal controls may divert management’s attention from other matters that are important to our business. Once we are no longer an “emerging growth company,” or a “smaller reporting company”, our auditors will be required to issue an attestation report on the effectiveness of our internal controls on an annual basis.
| 48 |
| Table of Contents |
In the course of preparing the financial statements that are included in this Annual Report on Form 10-K, management has determined that a material weakness exists within the internal controls over financial reporting. The material weakness identified relates to the lack of formal control documentation and consistent execution of control procedures, and the lack of a sufficient complement of personnel within the finance and accounting function with an appropriate degree of knowledge, experience and training. We also noted a material weakness related to logical security and privileged access in the area of information technology. We concluded that the material weaknesses in our internal control over financial reporting information technology occurred because, prior to our IPO, we were a private company and did not have the necessary business processes, systems, personnel, and related internal controls necessary to satisfy the accounting and financial reporting requirements of a public company.
In order to remediate the material weaknesses, we expect to enhance our formal documentation over internal control procedures and management controls infrastructure to allow for more consistent execution of control procedures and hire additional accounting, finance and information technology resources or consultants with public company experience.
We may not be able to fully remediate the identified material weakness until the steps described above have been completed and our internal controls have been operating effectively for a sufficient period of time. We believe we have already and will continue to make progress in our remediation plan but cannot assure you that we will be able to fully remediate the material weakness by such time. If the steps we take do not correct the material weakness in a timely manner, we will be unable to conclude that we maintain effective internal control over financial reporting. Accordingly, there could continue to be a reasonable possibility that a material misstatement of our financial statements would not be prevented or detected on a timely basis. We also may incur significant costs to execute various aspects of our remediation plan but cannot provide a reasonable estimate of such costs at this time.
In accordance with the provisions of the JOBS Act, we and our independent registered public accounting firm were not required to, and did not, perform an evaluation of our internal control over financial reporting as of December 31, 2023 nor any period subsequent in accordance with the provisions of the Sarbanes-Oxley Act. Accordingly, we cannot assure you that we have identified all material weaknesses. Material weaknesses may still exist when we report on the effectiveness of our internal control over financial reporting as required under Section 404 of the Sarbanes-Oxley Act.
In the future, it is possible that additional material weaknesses or significant deficiencies may be identified that we may be unable to remedy before the requisite deadline for these reports. Our ability to comply with the annual internal control reporting requirements will depend on the effectiveness of our financial reporting and data systems and controls across our company. Any weaknesses or deficiencies or any failure to implement new or improved controls, or difficulties encountered in the implementation or operation of these controls, could harm our operating results and cause us to fail to meet our financial reporting obligations, or result in material misstatements in our consolidated financial statements, which could adversely affect our business and reduce our stock price.
If we are unable to conclude on an ongoing basis that we have effective internal control over financial reporting in accordance with Section 404, our independent registered public accounting firm may not issue an unqualified opinion. If we are unable to conclude that we have effective internal control over financial reporting, investors could lose confidence in our reported financial information, which could have a material adverse effect on the trading price of our Common Stock. Failure to remedy any material weakness in our internal control over financial reporting, or to implement or maintain other effective control systems required of public companies, could also restrict our future access to the capital markets.
We could be subject to securities class action litigation.
In the past, securities class action litigation has often been brought against a company following a decline in the market price of its securities. This risk is especially relevant for us because biopharmaceutical companies have experienced significant stock price volatility in recent years. If we face such litigation, it could result in substantial costs and a diversion of management’s attention and resources, which could harm our business.
Our failure to meet Nasdaq’s continued listing requirements could result in a delisting of our Common Stock.
If we fail to satisfy the continued listing requirements of Nasdaq, such as the corporate governance requirements or the minimum closing bid price requirement, Nasdaq may take steps to delist our Common Stock. Such a delisting would likely have a negative effect on the price of our Common Stock and would impair your ability to sell or purchase our Common Stock when you wish to do so. In the event of a delisting, we can provide no assurance that any action taken by us to restore compliance with listing requirements would allow our Common Stock to become listed again, stabilize the market price or improve the liquidity of our Common Stock, prevent our Common Stock from dropping below the Nasdaq minimum bid price requirement or prevent future non-compliance with the listing requirements of Nasdaq.
| 49 |
| Table of Contents |
If securities or industry analysts do not publish research or publish inaccurate or unfavorable research about our business, our stock price and trading volume could decline.
The trading market for our Common Stock depends in part on the research and reports that securities or industry analysts publish about us or our business. Securities and industry analysts do not currently, and may never, publish research on our company. If no securities or industry analysts commence coverage of our company, the trading price for our stock would likely be negatively impacted. In the event securities or industry analysts initiate coverage, if one or more of the analysts who covers us downgrades our stock or publishes inaccurate or unfavorable research about our business, our stock price may decline. If one or more of these analysts ceases coverage of our company or fails to publish reports on us regularly, demand for our stock could decrease, which might cause our stock price and trading volume to decline.
Item 1B. Unresolved Staff Comments
None.
Item 1C. Cybersecurity
Cybersecurity Risk Management and Strategy
Due to the size of our company, we have not yet developed robust policies and processes for assessing, identifying, and managing material risk from cybersecurity threats. We have implemented access controls with respect to our systems, which we monitor regularly and audit annually. Our most sensitive data is stored in offline air-gapped devices. We currently rely heavily on products and services provided by third-party suppliers to operate certain critical business systems, including without limitation, cloud-based infrastructure, encryption and authentication technology, email, and other functions. We rely on third party providers and outsourced IT services to monitor and address cybersecurity related risks, including installing software for threat protection and malware. Such third party providers are tasked with notifying management of any material risks or cybersecurity concerns that they identify, which management then assesses and may bring to our board of directors to discuss if deemed necessary or appropriate. Based on the results of our risk assessments, if deemed necessary or appropriate, we take steps to re-design, implement, and maintain reasonable safeguards to minimize identified risks; reasonably address any identified gaps in existing safeguards; and regularly monitor the effectiveness of our safeguards.
We intend to work with outside counsel and third party service providers in the near term to further develop our expertise, processes and procedures with respect to cybersecurity protection and our response plan.
To date, we have not (to our knowledge) encountered cybersecurity challenges that have materially impaired our operations or financial standing. For additional information regarding risks from cybersecurity threats, please refer to Item 1A, “Risk Factors,” in this Report.
Governance
Our management team is primarily responsible for assessing and managing our strategic risk exposures, including material risks from cybersecurity threats, with assistance from third-party service providers. Management oversees our cybersecurity process on a day-to-day basis, including those described under the heading “Cybersecurity Risk Management and Strategy” above.
Our audit committee is tasked with general oversight of our risk management process, including risks from cybersecurity threats. Members of management provide periodic briefings to the audit committee of our board of directors regarding our cybersecurity risks and activities, including any recent cybersecurity incidents and related responses, cybersecurity systems testing, activities of third parties, and the like. In furtherance thereof, the committee is responsible for monitoring and assessing strategic risk exposure. Our audit committee provides regular updates to the board of directors on such reports.
Item 2. Properties
As of December 31, 2023, we lease five facilities in Pretoria, South Africa for office, production and laboratory space.
One lease commenced in October 2021 with the initial term set to expire in December 2030. This space is used for office, production and laboratory activities.
The second lease commenced in April 2023 with the initial term expired in March 2024. The Company plans to remain in this space under the monthly renewal terms of the agreement. This space is used for production and laboratory activities.
The third lease commenced in November 2023 with the initial term set to expire in October 2026. This space is used for laboratory activities.
| 50 |
| Table of Contents |
The fourth lease commenced with our acquisition of PET Labs Pharmaceuticals in October 2023 and has an initial term set to expire in March 2026 with automatic monthly extensions thereafter. This space is used for office and production activities.
The fifth lease commenced with our acquisition of PET Labs Pharmaceuticals in October 2023 and had an initial term which expired in December 2023 with automatic monthly extensions thereafter. This space is used for production activities.
We believe that our current facilities are sufficient to meet our current and near-term needs and that, should it be needed, suitable additional space will be available.
Item 3. Legal Proceedings
We are not party to any material legal matters or claims. In the future, we may become party to legal matters and claims arising in the ordinary course of business. We cannot predict the outcome of any such legal matters or claims, and despite the potential outcomes, the existence thereof may have an adverse impact on us because of defense and settlement costs, diversion of management resources and other factors.
Item 4. Mine Safety Disclosures
Not applicable.
| 51 |
| Table of Contents |
PART II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Market Information and Holders of Record
Our common stock has been listed on the Nasdaq Global Select Market under the symbol “ASPI” since November 10, 2022.
As of April 8, 2024, we had 30 record holders of record of our common stock. The actual number of shareholders is greater than this number of record holders and includes shareholders who are beneficial owners but whose shares are held in street name by brokers and other nominees. The number of holders of record also does not include shareholders whose shares may be held in trust by other entities.
Dividends
We have never declared or paid a cash dividend on our capital stock. We currently intend to retain any future earnings and do not expect to pay any dividends in the foreseeable future. Any future determinations to pay cash dividends will be made at the discretion of our board of directors, subject to applicable laws, and will depend on a number of factors, including our financial condition, results of operations, capital requirements, contractual restrictions, general business conditions, and any other factors that our board may deem relevant.
Securities Authorized for Issuance under Equity Compensation Plans
Information regarding the Securities Authorized for Issuance under our Equity Compensation Plans will be included in an amendment to this Annual Report in Form 10-K or incorporated by reference from our definitive proxy statement to be filed pursuant to Rule 14A.
Stock Performance Graph
As a smaller reporting company, we are not required to provide the information requested by this item pursuant to Item 201 of Regulation S-K.
Unregistered Sales of Equity Securities and Use of Proceeds
Unregistered sales of equity securities
None.
Use of proceeds from registered securities
On November 15, 2022, we completed our IPO, in which we issued and sold 1,250,000 shares of common stock, $0.01 par value per share at a price to the public of $4.00 per share. The offer and sale of the shares in the IPO was registered under the Securities Act pursuant to a registration statement on Form S-1 (File No. 333-267392), which was filed with the SEC on September 12, 2022 and amended subsequently and declared effective on November 9, 2022. The underwriter of the offering Revere Securities, LLC. The Form S-1 registered 2,057,500 shares of common stock held by selling stockholders. We did not receive proceeds from the sale of the shares by the selling stockholders.
We raised approximately $3.8 million in net proceeds after deducting underwriting discounts and commissions and other offering expenses of $1.2 million. No offering expenses were paid directly or indirectly to any of our directors of officers (or their associates) or persons owning ten percent or more of any class of our equity securities or to any other affiliates.
Through December 31, 2023, we have used all of the net proceeds from our IPO for matters described in our final IPO prospectus filed with the SEC on November 14, 2022, or our IPO prospectus. There has been no material change in the planned use of proceeds from our IPO, as described in our IPO prospectus.
Repurchases of equity securities by the issuer
None.
| 52 |
| Table of Contents |
Item 6. [Reserved]
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
The following discussion and analysis of financial condition and results of operations is provided to enhance the understanding of, and should be read in conjunction with Part I, Item I, “Business” and Item 8, ‘Financial Statements and Supplementary Data.” For information on risks and uncertainties related to our business that may make past performance not indicative of future results or cause actual results to differ materially from any forward-looking statements, see “Special Note Regarding Forward-Looking Statements,” and Part I, Item 1A, ‘Risk Factors.”
Overview
We are a development stage advanced materials company dedicated to the development of technology and processes that, if successful, will allow for the enrichment of natural isotopes into higher concentration products, which could be used in several industries. Our proprietary technology, the Aerodynamic Separation Process (“ASP technology”), originally developed by Klydon Proprietary Ltd (“Klydon”), is designed to enable the production of isotopes used in several industries. Our initial focus is on the production and commercialization of enriched Carbon-14 (“C-14”), Molybdenum-100 (“Mo-100”) and Silicon-28 (“Si-28”). We have commissioned an isotope enrichment plant for the enrichment of C-14 located in Pretoria, South Africa, which will be ready for production upon the final installation of essential components. We anticipate completion and commissioning of a multi-isotope enrichment plant in Pretoria, South Africa in mid-2024. In addition, we have started planning additional isotope enrichment plants. We believe the C-14 we may produce using the ASP technology could be used in the development of new pharmaceuticals and agrochemicals. We believe the Mo-100 we may produce using the ASP technology could have significant potential advantages for use in the preparation of nuclear imaging agents by radiopharmacies and others in the medical industry. We believe the Si-28 we may produce using the ASP technology may be used to create advanced semiconductors and in quantum computing. In addition, we are considering the future development of the ASP technology for the separation of Zinc-68, Xenon-129/136 for potential use in the healthcare end market, Germanium 70/72/74 for possible use in the semiconductor end market, and Chlorine -37 for potential use in the nuclear energy end market.
We are also developing Quantum Enrichment technology to produce enriched Ytterbium-176, Nickel-64, Lithium 6, Lithium7 and Uranium-235 (“U-235”). Quantum enrichment is an advanced isotope enrichment technique that is currently in development that uses lasers. We believe that the U-235 we may produce using quantum enrichment technology may be commercialized as a nuclear fuel component for use in the new generation of HALEU- fueled small modular reactors that are now under development for commercial and government uses.
On November 15, 2022, we completed an IPO of our common stock and issued and sold 1,250,000 shares of common stock at a public offering price of $4.00 per share, resulting in net proceeds of $3.8 million after deducting underwriting discounts and commissions and offering expenses.
In March 2023, we issued 3,164,557 shares of our common stock at a purchase price of $1.58 per share and warrants to purchase up to an aggregate of 3,164,557 shares of our common stock with an exercise price of $1.75 per share for gross proceeds of $5.0 million. We incurred $506,390 in cash issuance costs and issued warrants to purchase up to an aggregate of 221,519 shares of common stock with an exercise price of $1.975 per share to the placement agent with an initial fair value of $179,116.
In October 2023, the Company entered into Securities Purchase Agreements with certain institutional and other accredited investors and certain directors of the Company to issue and sell an aggregate of 9,952,510 shares of the Company’s common stock, for aggregate cash consideration of $9,129,461, as follows: (i) 8,459,093 shares to investors at a purchase price per share of $0.9105, (ii) 1,190,239 shares to investors at a purchase price per share of $0.9548, and (iii) 303,178 shares to directors at a purchase price per share of $0.96. The Company incurred issuance costs equivalent to 5% of the gross proceeds from new investors which was settled in stock through the issuance of 472,582 shares to the placement agent and additional cash issuance costs totaling $57,083.
Acquisition of 51% of PET Labs Pharmaceuticals
In October 2023, the Company entered into a Share Purchase Agreement with Nucleonics Imaging Proprietary Limited, a company incorporated in South Africa, to purchase 51% of the ordinary shares in Nucleonics’ wholly-owned subsidiary, Pet Labs Pharmaceuticals Proprietary Limited, a company incorporated in South Africa and dedicated to nuclear medicine and the science of radiopharmaceutical production.
| 53 |
| Table of Contents |
Per the Share Purchase Agreement, the Company has agreed to pay a total of $2,000,000 for the shares in two installments. The first installment of $500,000 was paid in November 2023. The remaining balance of $1,500,000 is due upon demand any time after October 31, 2024 and is expected to be paid in November 2024.
In March 2024, the Company’s wholly owned subsidiary Quantum Leap Energy received gross proceeds of $20,550,000 through the issuance of Convertible Promissory Notes with a stated interest rate of 6% for the first year and 8% thereafter. The maturity date of the Convertible Promissory Notes is March 7, 2029. The Convertible Promissory Notes automatically convert into common shares upon Quantum Leap Energy’s closing of an IPO or other qualifying public transaction at 80% of the share price taking into consideration a valuation cap.
In April 2024, the Company received approximately $5.5 million from the issuance of 3,164,557 shares of common stock upon the exercise of warrants.
Acquisition of Assets and Agreements with Klydon
To date, we have purchased certain assets of Molybdos Proprietary Limited, a South Africa company (Molybdos), and entered into a number of agreements with Klydon (Pty) Limited, a South Africa company (Klydon). Below is a summary of the key terms for our former licenses and other agreements with Klydon.
Acquisition of Molybdos Assets. On September 30, 2021, our subsidiary, ASP Isotopes South Africa (Proprietary) Limited (“ASP South Africa”), participated in and was declared the winner of a competitive auction process under Section 45 of the South Africa Consumer Protection Act, 2008 related to the sale and assignment of the assets of Molybdos (the “Molybdos Business Rescue Auction”). On October 12, 2021, ASP South Africa acquired the assets of Molybdos for ZAR 11,000,000 (which at the then current exchange rate was approximately $734,000), plus value added tax (VAT) levied by the government of South Africa at the rate of 15% and auctioneers’ commission at the rate of 10%.
Acquisition of Silicon-28 Plant Assets. On July 26, 2022, we acquired assets comprising a dormant Silicon-28 aerodynamic separation processing plant from Klydon for ZAR 6,000,000 (which at the then current exchange rate was approximately $364,000), which will be payable to Klydon on the later of 180 days of the acquisition and the date on which the assets generate any revenues of any nature.
Exclusive Mo-100 License (superseded and replaced by new license (see “Omnibus Klydon License” below)). On September 30, 2021, ASP South Africa, as licensee, entered into a license with Klydon, as licensor, pursuant to which ASP South Africa acquired from Klydon an exclusive license to use, subcontract and sublicense certain intellectual property rights relating to the ASP technology for the development and/or otherwise disposing of the ASP technology and production, distribution, marketing and or sale of Mo-100 isotope produced using the ASP technology (as amended on June 8, 2022, the “Mo-100 license”). The intellectual property rights granted to us through the Mo-100 license included all existing and/or future proprietary rights of Klydon relating to the ASP technology, whether or not such rights have been registered, including the copyright, designs, know-how, patents and trademarks (although Klydon currently has no such patents, patent applications or copyrights). The exclusive Mo-100 license was royalty-free, had a term of 999 years and was for the global development of the ASP Technology and production of the Mo-100 Isotope and global for the distribution, marketing and sale of the Mo-100 Isotope. No upfront or other payment was made or is owed in connection with the Mo-100 license. Klydon had the right to terminate the exclusivity of the Mo-100 license in the event that the licensee ceased carrying on activities of Mo-100 enrichment for a period longer than 24 consecutive months. Klydon had no other rights to terminate the Mo-100 license. Effective July 26, 2022, the parties agreed to terminate the Mo-100 license, which was superseded and replaced by a new license agreement (described under the heading “Omnibus Klydon License” below).
Exclusive U-235 License (superseded and replaced by new license (see “Omnibus Klydon License” below)). On January 25, 2022, ASP South Africa, as licensee, entered into a license with Klydon, as licensor, pursuant to which ASP South Africa acquired from Klydon an exclusive license to use, subcontract and sublicense certain intellectual property rights relating to the ASP technology for the development and/or otherwise disposing of the ASP technology and production, distribution, marketing and or sale of U-235 produced using the ASP (as so amended, the “U-235 license”). The intellectual property rights granted to us through the U-235 license included all existing and/or future proprietary rights of Klydon relating to the ASP technology, whether or not such rights have been registered, including the copyright, designs, know-how, patents and trademarks (although Klydon currently has no such patents, patent applications or copyrights). The exclusive U-235 license had a term of 999 years and was for the global development of the ASP technology and production of U-235 and global for the distribution, marketing and sale of U-235. In connection with the U-235 license we made an upfront payment of $100,000 and agreed to pay certain royalties (the greater of $50 per k.g. of U-235 and 10% of profits) and a 33% sublicensing revenue share of any cash consideration we may receive for any sublicenses we may grant. Klydon had the right to terminate the exclusivity of the U-235 license in the event that the licensee ceased carrying on activities of U-235 enrichment for a period longer than 24 consecutive months. Klydon had no other rights to terminate the U-235 license. Effective July 26, 2022, the parties agreed to terminate the U-235 license, which was superseded and replaced by a new license agreement (described under the heading “Omnibus Klydon License” below).
Omnibus Klydon License. On July 26, 2022, ASP Isotopes UK Ltd, as licensee, entered into a license agreement with Klydon, as licensor, pursuant to which ASP Isotopes UK Ltd acquired from Klydon an exclusive license to use, develop, modify, improve, subcontract and sublicense certain intellectual property rights relating to the ASP technology for the production, distribution, marketing and sale of all isotopes produced using the ASP technology (the “Klydon license agreement”). The intellectual property rights granted to us through the Klydon license agreement included all existing and/or future proprietary rights of Klydon relating to the ASP technology, whether or not such rights have been registered, including the copyright, designs, know-how, patents and trademarks (although Klydon currently has no such patents, patent applications or copyrights). The Klydon license agreement was royalty-free, had a term of 999 years and was worldwide for the development of the ASP technology and the distribution, marketing and sale of isotopes. Future production of isotopes is limited to member countries of the Nuclear Suppliers Group. In connection with the Klydon license agreement, we agreed to make an upfront payment of $100,000 (to be included within the payments we made under the Turnkey Contract (described below) and deferred payments of $300,000 over 24 months. Effective April 4, 2023, pursuant to the Acknowledgement of Debt Agreement described below, we acquired the ASP technology, among other things, from Klydon, and the Klydon license agreement is no longer in effect.
| 54 |
| Table of Contents |
Turnkey Contract. On November 1, 2021, ASP South Africa and Klydon, as the contractor, entered into a contract under which Klydon has been appointed to supply to ASP South Africa a complete turnkey isotope enrichment plant (the “Turnkey Contract”). The activities to be undertaken or performed by Klydon include: taking control of the assets acquired in the Molybdos Business Rescue Auction; the design of an isotope enrichment facility; the supply of components, equipment and labor required for the construction; the installation, testing and commissioning of the isotope enrichment plant; securing all required approvals, regulatory authorizations and other required consents for the operation of the plant; providing training to local ASP Isotopes South Africa (Proprietary) Limited personnel to enable them to operate the plant going forward; and providing warranties in relation to the performance targets of the plant which are required to be met. Klydon was responsible for liaising with the relevant South African authorities, including the South African Non Proliferation Council, the Nuclear Suppliers Group and International Atomic Energy Agency to ensure that the Turnkey Contract and the isotope enrichment plant are compliant with international laws and guidelines.
Acknowledgement of Debt Agreement. Klydon performed a portion of the services required under the Turnkey Contract described above; however, services were incomplete and many of the services were not completed within the time frame required. As a result, Klydon and ASP South Africa entered into an Acknowledgement of Debt Agreement dated November 30, 2022, whereby Klydon (i) agreed to pledge its assets (the “Pledged Assets”) to ASP South Africa to secure its performance of the Turnkey Contract by December 31, 2022, and (ii) acknowledged that ASP South Africa would suffer damages in the amount of $6,050,000 (“Damage Amount”) should it fail to perform. Under the Acknowledgement of Debt Agreement, the Pledged Assets would serve as collateral for Klydon’s obligation to pay the Damage Amount should Klydon fail to perform. In connection therewith, also on November 30, 2022, ASP South Africa and Klydon entered into a Deed of Security Agreement whereby, if Klydon failed to complete its obligations under the Turnkey Contract by December 31, 2022, all of Klydon’s rights of any nature to and interests of any nature in the Pledged Assets would be transferred to ASP South Africa. Klydon failed to complete its obligations under the Turnkey Contract by December 31, 2022.
On April 4, 2023, the Company perfected its interests in the assets under the Acknowledgement of Debt Agreement, pursuant to which the Company acquired the Pledged Assets, including certain intellectual property, from Klydon and settled all amounts due to Klydon, including the ZAR 6,000,000 for the acquisition of the Silicon-28 plant assets.
Other Commercial Agreements
Below is a summary of the key terms of our other commercial agreements.
Lease for Molybdenum Processing Plant. On October 12, 2021, ASP South Africa entered into an agreement of lease with the landlord of the facility located at 33 Eland Street, Koedoespoort Industrial, Pretoria where we operate our Molybdenum processing plant where gaseous Molybdenum compound will be treated (which process comprises several stages of compression and expansion during which the product is purified). The term of the lease ends on December 31, 2030.
Lease for additional production space. On April 1, 2023, ASP South Africa entered into an agreement of lease with the landlord of facility located in Pretoria where we plan to perform production activities. The initial term of the lease ended on March 31, 2024. The Company intends to maintain the monthly extensions allowed in the lease.
Lease for additional laboratory space. On November 1, 2023, ASP South Africa entered into an agreement of lease with the landlord of the facility located in Pretoria where we perform research and development activities. The term of the lease ends on October 30, 2026.
Lease for PET Labs Pharmaceutical operations. Commencing with our acquisition of PET Labs Pharmaceuticals in October 2023, this facility has an initial term set to expire in March 2026 with automatic monthly extensions thereafter. This space is used for office and production activities.
Lease for additional PET Labs Pharmaceutical operations. Commencing with our acquisition of PET Labs Pharmaceuticals in October 2023, this facility had an initial term which expired in December 2023 and is currently under automatic monthly extensions. This space is used for production activities.
Political Risk Insurance Policy with Optio Group. On October 25, 2021, ASP Guernsey entered into a contract of insurance to cover against political risk and expropriation, to off-set the risk of events detrimental to the company occurring in the Republic of South Africa for a period of three years. The insurer is Optio Group Limited which is 100% underwritten by one or more syndicates at Lloyd’s of London. The specific risks covered in the policy are: (i) permanent and total abandonment of operations, (ii) deprivation of assets or shareholding, (iii) physical damage due to political violence, (iv) non-transfer or inconvertibility, (v) business interruption, (vi) non-honouring of arbitration award, and (vii) crisis management support. The limit of cover is equal to or in excess of the projected amount of investment required to complete the initial stage of the first planned Molybdenum enrichment plant. The limit of cover is capable of being increased and extended by mutual agreement with the insurer.
| 55 |
| Table of Contents |
Components of Results of Operations
Revenue
Effective with the acquisition of 51% of PET Labs Pharmaceuticals, the Company recognizes revenue from the sale of nuclear medical doses for PET scanning.
Cost of Goods Sold
Cost of goods sold associated with the sale of nuclear medical doses for PET scanning consist of labor, delivery and materials.
Operating Expenses
Our operating expenses consist of (i) research and development expenses and (ii) selling, general and administrative expenses.
Research and Development
Our research and development expenses consist primarily of direct and indirect costs incurred in connection with the development activities for our future isotopes.
Direct costs include:
| · | external research and development expenses; and |
|
|
|
| · | costs related to designing the development processes of isotope production. |
Indirect costs include:
| · | personnel-related costs, which include salaries, payroll taxes, employee benefits, and other employee-related costs, including stock-based compensation, for personnel engaged in research and development functions; and |
|
|
|
| · | facilities and other various expenses. |
Research and development expenses are recognized as incurred and payments made prior to the receipt of goods or services to be used in research and development are capitalized until the goods or services are received.
As described above, Klydon charged us for expenses associated with these research and development functions under the Turnkey Contract. We expect that our research and development expenses will increase substantially for the foreseeable future as we continue the development of our future isotopes. We cannot determine with certainty the timing of initiation, the duration or the completion costs of development activities. Actual development timelines, the probability of success and development costs can differ materially from expectations.
We will need to raise substantial additional capital in the future. In addition, we cannot forecast which future isotopes may be subject to future collaborations, when such arrangements will be secured, if at all, and to what degree such arrangements would affect our development plans and capital requirements.
Our research and development expenses may vary significantly based on a variety of factors, such as:
| · | the scope, rate of progress, expense and results of our development activities; |
|
|
|
| · | the phase of development of our future isotopes; |
|
|
|
| · | the timing, receipt, and terms of any approvals from applicable regulatory authorities including the FDA and foreign regulatory authorities; |
|
|
|
| · | significant and changing government regulation and regulatory guidance; |
|
|
|
| · | the cost and timing of designing the development processes of isotope production; |
|
|
|
| · | the extent to which we establish additional strategic collaborations or other arrangements; and |
|
|
|
| · | the impact of any business interruptions to our operations or to those of the third parties with whom we work. |
A change in the outcome of any of these variables with respect to the development of any of our future isotopes could significantly change the costs and timing associated with the development of that future isotope.
| 56 |
| Table of Contents |
Selling, General and Administrative
Selling, general and administrative expenses consist primarily of personnel-related costs, which include salaries, payroll taxes, employee benefits, and other employee-related costs, including stock-based compensation, for personnel in executive, sales, finance and other administrative functions. Other significant costs include legal fees relating to corporate matters, professional fees for accounting and consulting services and facility-related costs.
We expect that our ongoing selling, general and administrative expenses will increase substantially for the foreseeable future to support our increased research and development activities and increased costs of operating as a public company and in building our internal resources. These increased costs will include increased expenses related to audit, legal, regulatory and tax-related services associated with maintaining compliance with exchange listing and SEC requirements, director and officer insurance premiums and investor and public relations costs associated with operating as a public company.
Results of Operations
Comparison of the Years Ended December 31, 2023 and 2022
The following table summarizes our results of operations for the years ended December 31, 2023 and 2022:
|
| Year Ended December 31, 2023 |
|
| Year Ended December 31, 2022 |
| ||
Revenue |
| $ | 433,026 |
|
| $ | - |
|
Cost of goods sold |
|
| 294,056 |
|
|
| - |
|
Gross margin |
|
| 138,970 |
|
|
| - |
|
|
|
|
|
|
|
|
|
|
Operating expenses: |
|
|
|
|
|
|
|
|
Research and development |
|
| 764,581 |
|
|
| 1,273,536 |
|
Selling, general and administrative |
|
| 15,416,388 |
|
|
| 3,825,512 |
|
Total operating expenses |
|
| 16,180,969 |
|
|
| 5,099,048 |
|
Other (expense) income: |
|
|
|
|
|
|
|
|
Foreign exchange transaction gain |
|
| 45,753 |
|
|
| - |
|
Change in fair value of share liability |
|
| (194,540 | ) |
|
| 150,527 |
|
Interest expense |
|
| (118,547 | ) |
|
| - |
|
Interest income |
|
| 9,074 |
|
|
| 3,382 |
|
Total other (expense) income |
|
| (258,260 | ) |
|
| 153,909 |
|
Loss before income tax expense |
| $ | (16,300,259 | ) |
| $ | (4,945,139 | ) |
Revenue and Cost of Goods Sold
Effective with the acquisition of 51% of PET Labs Pharmaceuticals, the Company has recognized revenue from the sale of nuclear medical doses for PET scanning for the two month period since the acquisition was effective on October 31, 2023 and December 31, 2023. In addition, the Company has recognized the related cost of goods sold, operating expenses and other income and expenses for the same period.
Research and Development Expenses
The following table summarizes our research and development expenses for the years ended December 31, 2023 and 2022:
|
| Year Ended December 31, |
|
| Year Ended December 31, |
| ||
|
| 2023 |
|
| 2022 |
| ||
Direct costs: |
|
|
|
|
|
| ||
Mo-100 |
| $ | - |
|
| $ | 6,645 |
|
Indirect costs: |
|
|
|
|
|
|
|
|
Personnel-related costs |
|
| 495,034 |
|
|
| 429,270 |
|
License fees |
|
| - |
|
|
| 495,503 |
|
Consulting, facility and other expenses |
|
| 269,547 |
|
|
| 342,118 |
|
Total research and development expenses |
| $ | 764,581 |
|
| $ | 1,273,536 |
|
| 57 |
| Table of Contents |
Research and development expenses were $764,581 for the year ended December 31, 2023. These expenses include $495,034 of personnel-related costs, including $364,924 in stock-based compensation, and $269,547 in consulting, facility and other expenses.
Research and development expenses were $1,273,536 for the year ended December 31, 2022. These expenses include $6,645 in consulting expenses related to advancing development activities for Mo-100, $429,270 of personnel-related costs, including $201,270 in stock-based compensation, $495,503 in license fees and $342,118 in consulting, facility and other expenses.
The increase in stock-based compensation is due to a full year of expense in 2023 versus a partial year in 2022 since a majority of the awards were made in the second half of 2022. The decrease in consulting, facility and other expenses is mainly due to lower consulting costs in 2023 as the Company focused its activities in 2023 on completing the construction of the plant.
Selling, General and Administrative Expenses
Selling, general and administrative expenses were $15,416,388 for the year ended December 31, 2023. These expenses include $10,422,994 of personnel-related costs, including $8,378,875 in stock-based compensation, $2,747,486 of professional services and legal related fees and $2,245,909 in facility and other corporate expenses.
Selling, general and administrative expenses were $3,825,512 for the year ended December 31, 2022. These expenses include $560,789 of personnel-related costs, $1,798,043 in stock-based compensation, $1,010,187 of professional services and legal related fees and $456,493 in facility and other corporate expenses.
The increase in stock-based compensation is due to a full year of expense in 2023 versus a partial year in 2022 since a majority of the awards were made in the second half of 2022. The increase in professional services and legal related fees and in facility and other expenses is mainly due to increased costs related to being a public entity and the expansion of our operations in 2023.
Other Income and Expense
Other expense for the year ended December 31, 2023 was $258,260, which includes a $194,540 change in the fair value of the share liability related to the shares issuable to a placement agent and other consultants and interest expense of $118,547.
Other income for the year ended December 31, 2022 was $153,909, which includes a $150,527 change in the fair value of the share liability related to the shares issuable to a placement agent.
Liquidity and Capital Resources
Sources of Liquidity
We have incurred net losses and negative cash flows from operations since our inception, and we expect to continue to incur significant and increasing net losses for the foreseeable future. We have principally financed our operations to date through the issuance of our common stock, including our IPO. On March 7, 2024, the Company’s wholly owned subsidiary Quantum Leap Energy received gross proceeds of $20,550,000 through the issuance of Convertible Promissory Notes with a stated interest rate of 6% for the first year and 8% thereafter. The maturity date of the Convertible Promissory Notes is March 7, 2029. The Convertible Promissory Notes automatically convert into common shares upon Quantum Leap Energy’s closing of an IPO or other qualified public transaction at 80% of the share price taking into consideration a valuation cap. On April 9, 2024, the Company received approximately $5,500,000 from the issuance of 3,164,557 shares of common stock upon the exercise of warrants.
As of December 31, 2023, we had cash of $7.9 million. We do not have any isotopes approved for sale, we have not generated any revenue from the sale of isotopes, and our ability to generate product revenue from the sale of isotopes sufficient to achieve profitability will depend on the successful development and eventual commercialization of one or more of our current or future isotopes.
Effective with the acquisition of 51% of PET Labs Pharmaceuticals on October 31, 2023, we have begun to recognize revenue from the sale of nuclear medical doses for PET scanning in South Africa. Our ability to generate product revenue from the sale of nuclear medical doses for PET scanning sufficient to achieve profitability will depend on the successful expansion of production capabilities and commercialization of the results of that expansion.
| 58 |
| Table of Contents |
Future Funding Requirements
Based on our current operating plan, we estimate that our existing cash, will not be sufficient to fund our operating expenses and capital expenditure requirements through at least the next 12 months from the date the financial statements are issued. However, our forecast of the period of time through which our financial resources will be adequate to support our operations is a forward-looking statement that involves risks and uncertainties, and actual results could vary materially. We have based this estimate on assumptions that may prove to be wrong, and we could deplete our capital resources sooner than we expect. Additionally, the process of developing isotopes is costly, and the timing of progress and expenses in these development activities is uncertain.
Our future capital requirements will depend on many factors, including:
| · | the type, number, scope, progress, expansions, results, costs and timing of, our development activities for our future isotopes; |
|
|
|
| · | the outcome, timing and costs of regulatory review of our future isotopes; |
|
|
|
| · | the costs and timing of manufacturing for our future isotopes; |
|
|
|
| · | our efforts to enhance operational systems and hire additional personnel to satisfy our obligations as a public company, including enhanced internal controls over financial reporting; |
|
|
|
| · | the costs associated with hiring additional personnel and consultants as our preclinical and clinical activities increase; |
|
|
|
| · | the costs and timing of establishing or securing sales and marketing and distribution capabilities, whether alone or with third parties, to commercialize future isotopes for which we may obtain regulatory approval, if any; |
|
|
|
| · | our ability to achieve sufficient market acceptance, coverage and adequate reimbursement from third-party payors and adequate market share and revenue for any approved products; |
|
|
|
| · | the terms and timing of establishing and maintaining collaborations, licenses and other similar arrangements; |
|
|
|
| · | the costs of obtaining, expanding, maintaining and enforcing our patent and other intellectual property rights; and |
|
|
|
| · | costs associated with any products or technologies that we may in-license or acquire. |
Developing isotopes is a time-consuming, expensive and uncertain process that takes years to complete, and we may never achieve the necessary results required or obtain applicable regulatory approval for any isotopes or generate revenue from the sale of any future isotopes (assuming applicable regulatory approval is received). In addition, our future isotopes (assuming applicable regulatory approval is received) may not achieve commercial success. Our commercial revenues, if any, will be derived from sales of isotopes that we do not expect to be commercially available in substantial quantities until at least 2024. If we receive permits and licenses to enrich U-235 (which in itself is highly uncertain), we do not expect U-235 to be commercially available for at least several years, if ever. As a result, we may need substantial additional financing to support our continuing operations and further the development of and commercialization of our future isotopes.
Expansion of the production and distribution of nuclear medical doses for PET scanning is a time-consuming, expensive and uncertain process that may take years to complete. As a result, we may need substantial additional financing to support our continuing operations and further the development of and commercialization of future nuclear medical doses for PET scanning.
Until such time as we can generate significant revenue from sales of our future isotopes or nuclear medical doses for PET scanning, if ever, we expect to finance our cash needs through public or private equity or debt financings or other capital sources, including potential collaborations, licenses and other similar arrangements. However, we may be unable to raise additional funds or enter into such other arrangements when needed on favorable terms or at all. Our ability to raise additional funds may be adversely impacted by potential worsening global economic conditions and the recent disruptions to, and volatility in, the credit and financial markets in the United States and worldwide resulting severely diminished liquidity and credit availability, increased interest rates, inflationary pressures, declines in consumer confidence, declines in economic growth, increases in unemployment rates and uncertainty about economic stability. The financial markets and the global economy may also be adversely affected by the current or anticipated impact of military conflict. To the extent that we raise additional capital through the sale of equity or convertible debt securities, the ownership interest of our stockholders will be or could be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect the rights of our common stockholders. Debt financing and equity financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. If we raise funds through collaborations, or other similar arrangements with third parties, we may have to relinquish valuable rights to our future isotopes, future revenue streams or research programs or may have to grant licenses on terms that may not be favorable to us and/or may reduce the value of our common stock. If we are unable to raise additional funds through equity or debt financings when needed, we may be required to delay, limit, reduce or terminate our product development or future commercialization efforts or grant rights to develop and market our future isotopes even if we would otherwise prefer to develop and market such isotopes ourselves.
| 59 |
| Table of Contents |
Cash Flows
The following table summarizes our sources and uses of cash for each of the periods presented:
|
| Year Ended December 31, |
|
| Year Ended December 31, |
| ||
|
| 2023 |
|
| 2022 |
| ||
Net cash provided by (used in): |
|
|
|
|
|
| ||
Operating activities |
| $ | (5,412,392 | ) |
| $ | (2,939,893 | ) |
Investing activities |
|
| (2,453,191 | ) |
|
| (4,473,164 | ) |
Financing activities |
|
| 13,385,491 |
|
|
| 6,641,052 |
|
Net increase (decrease) in cash and cash equivalents |
| $ | 5,519,908 |
|
| $ | (772,005 | ) |
Operating Activities
Net cash used in operating activities was $5,412,392 for the year ended December 31, 2023 and was primarily due to our net loss of $16,294,126, adjusted for stock-based compensation expense of $8,743,799, amortization of right-of-use asset of $104,528, issuance of common stock to a consultant with a fair value of $669,700, change in fair value of share liability of $194,540, and a $1,176,383 change in our operating assets and liabilities.
Net cash used in operating activities was $2,939,893 for the year ended December 31, 2022 and was primarily due to our net loss of $4,945,139, adjusted for stock-based compensation expense of $1,999,313, amortization of right-of-use asset of $72,570, issuance of common stock to a consultant with a fair value of $50,000 and change in fair value of share liability of $150,527, partially offset by a $33,890 change in our operating assets and liabilities.
Investing Activities
Net cash used in investing activities was $2,453,191 for the year ended December 31, 2023 and was comprised of the purchase of machinery and equipment and construction in progress totaling $2,331,343 and $121,848 for the acquisition of PET Labs net of cash acquired.
Net cash used in investing activities was $4,473,164 for the year ended December 31, 2022 and was comprised of construction in progress.
Financing Activities
Net cash provided by financing activities was $13,385,491 for the year ended December 31, 2023 and was comprised primarily of net proceeds of $13,566,022 from the sale and issuance of 13,117,067 shares of our common stock.
Net cash provided by financing activities was $6,641,052 for the year ended December 31, 2022 and was comprised primarily of net proceeds of $3,790,504 from the sale and issuance of 1,250,000 shares of our common stock in our IPO, net proceeds of $2,863,595 from the sale and issuance of 1,559,780 shares of our common stock prior to our IPO and the repayment of notes payable of $13,046.
Contractual Obligations and Commitments
We lease our main facility in Pretoria, South Africa under a lease with a base monthly rent payment of approximately $8,000 with a term expiring on December 31, 2030. We also lease additional space on a short term basis in Pretoria, South Africa under a lease with a base monthly rent payment of approximately $12,000 with an initial term that expired on March 31, 2024 and the Company is continuing to occupy that space under the monthly extensions. We also lease additional space in Pretoria, South Africa under a lease with a base monthly rent payment of approximately $2,000 with a term expiring on October 30, 2026.
PET Labs Pharmaceuticals operates in a facility in Pretoria, South Africa is under a lease with a base monthly rent payment of approximately $28,000 with a term expiring on March 30. 2026 with automatic monthly extension afterwards. PET Labs Pharmaceuticals also rents space at a local hospital in Pretoria, South Africa for which there was a lease with a base monthly rent payment of approximately $5,000 which expired on December 31, 2023 and is currently in automatic monthly extensions.
In addition, we enter into contracts in the normal course of business with vendors for services and products for operating purposes. These contracts do not contain any minimum purchase commitments and generally provide for termination after a notice period and, therefore, are not considered long-term contractual obligations. Payments due upon cancellation consist only of payments for services provided and expenses incurred up to the date of cancellation.
| 60 |
| Table of Contents |
Off-balance Sheet Arrangements
We did not have during the periods presented, and we do not currently have, any off-balance sheet arrangements, as defined in the rules and regulations of the SEC.
Critical Accounting Policies and Significant Judgments and Estimates
See Note 2 to our consolidated financial statements which discusses new accounting pronouncements.
Item 7A. Quantitative and Qualitative Disclosures About Market Risk
Interest Rate Risk
As of December 31, 2023 and 2022, our cash consists of cash in readily available checking accounts. We do not hold any short-term investments. As a result, the fair value of our portfolio is relatively insensitive to interest rate changes. As of December 31, 2023 and 2022, we had no bank debt outstanding and are therefore not exposed to interest rate risk with respect to debt. We believe a hypothetical 100 basis point increase or decrease in interest rates during the period presented would not have had a material impact on our financial results.
Foreign Currency Exchange Rate Risk
Our expenses are generally denominated in U.S. dollars but our operations are currently primarily located outside the United States and we have entered into a number of contracts with vendors that are denominated in foreign currencies. We are subject to foreign currency transaction gains or losses on our contracts denominated in foreign currencies. To date, foreign currency transaction gains and losses have not been material to our financial statements, and we have not had a formal hedging program with respect to foreign currency. We believe a hypothetical 100 basis point increase or decrease in exchange rates during the period presented would not have had a material impact on our financial results.
Effects of Inflation
Inflation generally affects us by increasing our cost of labor and research and development costs. We do not believe that inflation and changing prices had a significant impact on our results of operations for the period presented herein.
| 61 |
| Table of Contents |
Item 8. Financial Statements and Supplementary Data
ASP Isotopes Inc.
Index to Consolidated Financial Statements
| 62 |
| Table of Contents |
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Stockholders of
ASP Isotopes Inc
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of ASP Isotopes Inc. and Subsidiaries (the “Company”) as of December 31, 2023 and 2022, and the related consolidated statements of operations and comprehensive loss, changes in stockholders’ equity, and cash flows for each of the years then ended, and the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the consolidated financial position of the Company as of December 31, 2023 and 2022, and the consolidated results of their operations and their cash flows for each of the years then ended, in conformity with accounting principles generally accepted in the United States of America.
Going Concern
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the financial statements, the Company has incurred recurring operating losses and negative cash flows from operating activities that raise substantial doubt about its ability to continue as a going concern. Management’s plans in regard to these matters are also described in Note 1. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ EisnerAmper LLP
We have served as the Company’s auditor since 2022.
April 10, 2024
| 63 |
| Table of Contents |
ASP Isotopes Inc.
Consolidated Balance Sheets
|
| December 31, |
| |||||
|
| 2023 |
|
| 2022 |
| ||
Assets |
|
|
|
|
|
| ||
Current assets: |
|
|
|
|
|
| ||
Cash |
| $ |
|
| $ |
| ||
Accounts receivable |
|
|
|
|
|
| ||
Receivable from noncontrolling interests |
|
|
|
|
|
| ||
Prepaid expenses and other current assets |
|
|
|
|
|
| ||
Total current assets |
|
|
|
|
|
| ||
Property and equipment, net |
|
|
|
|
|
| ||
Operating lease right-of-use assets, net |
|
|
|
|
|
| ||
Goodwill |
|
|
|
|
|
| ||
Other noncurrent assets |
|
|
|
|
|
| ||
Total assets |
| $ |
|
| $ |
| ||
|
|
|
|
|
|
|
|
|
Liabilities and stockholders’ equity |
|
|
|
|
|
|
|
|
Current liabilities: |
|
|
|
|
|
|
|
|
Accounts payable |
| $ |
|
| $ |
| ||
Accrued expenses |
|
|
|
|
|
| ||
Notes payable |
|
|
|
|
|
| ||
Finance lease liabilities – current |
|
|
|
|
|
| ||
Operating lease liabilities – current |
|
|
|
|
|
| ||
Deferred revenue |
|
|
|
|
|
| ||
Other current liabilities |
|
|
|
|
|
| ||
Share liability |
|
|
|
|
|
| ||
Total current liabilities |
|
|
|
|
|
| ||
Deferred tax liabilities |
|
|
|
|
|
| ||
Finance lease liabilities – noncurrent |
|
|
|
|
|
| ||
Operating lease liabilities – noncurrent |
|
|
|
|
|
| ||
Other liabilities |
|
|
|
|
|
| ||
Total liabilities |
|
|
|
|
|
| ||
Commitments and contingencies (Note 8) |
|
|
|
|
|
|
|
|
Stockholders’ equity |
|
|
|
|
|
|
|
|
Preferred stock, $ |
|
|
|
|
|
| ||
Common stock, $ |
|
|
|
|
|
| ||
Additional paid-in capital |
|
|
|
|
|
| ||
Accumulated deficit |
|
| ( | ) |
|
| ( | ) |
Accumulated other comprehensive (loss) income |
|
| ( | ) |
|
|
| |
Total ASP Isotopes stockholders’ equity |
|
|
|
|
|
| ||
Noncontrolling interests |
|
|
|
|
|
| ||
Total stockholders’ equity |
|
|
|
|
|
| ||
Total liabilities and stockholders’ equity |
| $ |
|
| $ |
| ||
The accompanying notes are an integral part of these consolidated financial statements.
| 64 |
| Table of Contents |
ASP Isotopes Inc.
Consolidated Statements of Operations and Comprehensive Loss
|
| Year Ended December 31, 2023 |
|
| Year Ended December 31, 2022 |
| ||
|
|
|
|
|
|
| ||
Revenue |
| $ |
|
| $ |
| ||
Cost of goods sold |
|
|
|
|
|
| ||
Gross profit |
|
|
|
|
|
| ||
Operating expenses: |
|
|
|
|
|
|
|
|
Research and development |
|
|
|
|
|
| ||
Selling, general and administrative |
|
|
|
|
|
| ||
Total operating expenses |
|
|
|
|
|
| ||
Loss from operations |
|
| ( | ) |
|
| ( | ) |
Other income (expense): |
|
|
|
|
|
|
|
|
Foreign exchange transaction gain |
|
|
|
|
|
| ||
Change in fair value of share liability |
|
| ( | ) |
|
|
| |
Interest income |
|
|
|
|
|
| ||
Interest expense |
|
| ( | ) |
|
|
| |
Total other (expense) income |
|
| ( | ) |
|
|
| |
Loss before income tax expense |
|
| ( | ) |
|
| ( | ) |
Income tax provision |
|
|
|
|
|
| ||
Net loss before allocation to noncontrolling interests |
|
| ( | ) |
|
| ( | ) |
Less: Net loss attributable to noncontrolling interests |
|
| ( | ) |
|
|
| |
Net loss attributable to ASP Isotopes Inc. shareholders |
| $ | ( | ) |
| $ | ( | ) |
Net loss per share attributable to ASP Isotopes Inc. shareholders, basic and diluted |
| $ | ( | ) |
| $ | ( | ) |
Weighted average shares of common stock outstanding, basic and diluted |
|
|
|
|
|
| ||
|
|
|
|
|
|
|
|
|
Comprehensive loss: |
|
|
|
|
|
|
|
|
Net loss before allocation to noncontrolling interests |
| $ | ( | ) |
| $ | ( | ) |
Foreign currency translation |
|
| ( | ) |
|
|
| |
Total comprehensive loss before allocation to noncontrolling interests |
| ( | ) |
| ( | ) | ||
Less: Comprehensive loss attributable to noncontrolling interests |
|
|
|
|
|
| ||
Comprehensive loss attributable to ASP Isotopes Inc. |
| $ | ( | ) |
| $ | ( | ) |
The accompanying notes are an integral part of these consolidated financial statements.
| 65 |
| Table of Contents |
ASP Isotopes Inc.
Consolidated Statements of Changes in Stockholders’ Equity
|
| Common Stock |
|
| Additional Paid-in |
|
| Accumulated Other Comprehensive |
|
| Accumulated |
|
| Noncontrolling |
|
| Total Stockholders’ |
| ||||||||||
|
| Shares |
|
| Amount |
|
| Capital |
|
| Income |
|
| Deficit |
|
| Interests |
|
| Equity |
| |||||||
Balance as of December 31, 2021 |
|
|
|
| $ |
|
| $ |
|
| $ |
|
| $ | ( | ) |
| $ |
|
| $ |
| ||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuance of common stock, net of issuance costs of $380,747 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||
Issuance of common stock in connection with initial public offering, net of issuance costs of $1,209,496 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||
Issuance of common stock upon exercise of warrants |
|
|
|
|
|
|
|
| ( | ) |
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Issuance of restricted shares |
|
|
|
|
|
|
|
| ( | ) |
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Stock-based compensation |
|
| — |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Foreign currency translation |
|
| — |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Net loss |
|
| — |
|
|
|
|
|
|
|
|
|
|
|
| ( | ) |
|
|
|
|
| ( | ) | ||||
Balance as of December 31, 2022 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| ( | ) |
|
|
|
|
|
| ||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuance of common stock, net of issuance costs of $563,473 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||
Issuance of common stock for non-cash issuance costs |
|
|
|
|
|
|
|
| ( | ) |
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Issuance of common stock to settle share liability |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||
Settlement of liability with related parties |
|
| — |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Settlement of liability with consultants |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||
Cancellation of common stock received in exchange for issuance of convertible preferred stock in subsidiary |
|
| ( | ) |
|
| ( | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||
Issuance of restricted shares |
|
|
|
|
|
|
|
| ( | ) |
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Stock-based compensation |
|
| — |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Noncontrolling interest in ASP Rentals |
|
| — |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Acquisition of PET Labs |
|
| — |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ||||||
Foreign currency translation |
|
| — |
|
|
|
|
|
|
|
|
| ( | ) |
|
|
|
|
|
|
|
| ( | ) | ||||
Net loss |
|
| — |
|
|
|
|
|
|
|
|
|
|
|
| ( | ) |
|
| ( | ) |
|
| ( | ) | |||
Balance as of December 31, 2023 |
|
|
|
| $ |
|
| $ |
|
| $ | ( | ) |
| $ | ( | ) |
| $ |
|
| $ |
| |||||
The accompanying notes are an integral part of these consolidated financial statements.
| 66 |
| Table of Contents |
ASP Isotopes Inc.
Consolidated Statements of Cash Flows
|
| Year Ended December 31, 2023 |
|
| Year Ended December 31, 2022 |
| ||
Cash flows from Operating activities |
|
|
|
|
|
| ||
Net loss |
| $ | ( | ) |
| $ | ( | ) |
Adjustments to reconcile net loss to cash used in operating activities: |
|
|
|
|
|
|
|
|
Foreign exchange transaction gain from intercompany |
|
| ( | ) |
|
|
| |
Depreciation |
|
|
|
|
|
| ||
Stock-based compensation |
|
|
|
|
|
| ||
Issuance of common stock to consultant |
|
|
|
|
|
| ||
Change in fair value of share liability |
|
|
|
|
| ( | ) | |
Change in right-of-use lease assets |
|
|
|
|
|
| ||
Change in deferred tax liability |
|
|
|
|
|
| ||
Changes in operating assets and liabilities, net of acquisition amounts: |
|
|
|
|
|
|
|
|
Accounts receivable |
|
|
|
|
|
| ||
Prepaid expenses and other current assets |
|
| ( | ) |
|
| ( | ) |
Other noncurrent assets |
|
| ( | ) |
|
| ( | ) |
Accounts payable |
|
| ( | ) |
|
|
| |
Accrued expenses |
|
|
|
|
|
| ||
Operating lease liability |
|
| ( | ) |
|
| ( | ) |
Tax liability current |
|
| ( | ) |
|
|
| |
Deferred revenue |
|
|
|
|
|
| ||
Other current liabilities |
|
|
|
|
|
| ||
Net cash used in operating activities |
|
| ( | ) |
|
| ( | ) |
Cash flows from investing activities |
|
|
|
|
|
|
|
|
Purchases of property and equipment |
|
| ( | ) |
|
| ( | ) |
Cash paid for acquisition of business, net of cash acquired |
|
| ( | ) |
|
|
| |
Net cash used in investing activities |
|
| ( | ) |
|
| ( | ) |
Cash flows from financing activities |
|
|
|
|
|
|
|
|
Proceeds from issuance of common stock |
|
|
|
|
|
| ||
Common stock issuance costs |
|
| ( | ) |
|
| ( | ) |
Proceeds from issuance of notes payable |
|
|
|
|
|
| ||
Payments of notes payable |
|
| ( | ) |
|
|
| |
Payment of bank loan |
|
| ( | ) |
|
|
|
|
Payment of principal portion of finance leases |
|
| ( | ) |
|
| ( | ) |
Net cash provided by financing activities |
|
|
|
|
|
| ||
Net change in cash |
|
|
|
|
| ( | ) | |
Effect of exchange rate changes on cash |
|
| ( | ) |
|
|
| |
Cash – beginning of year |
|
|
|
|
|
| ||
Cash – end of year |
| $ |
|
| $ |
| ||
|
|
|
|
|
|
|
|
|
Supplemental disclosures of non-cash investing and financing activities: |
|
|
|
|
|
|
|
|
Issuance of common stock in lieu of commissions |
| $ |
|
| $ |
| ||
Settlement of liabilities with related party |
| $ |
|
| $ | — |
| |
Share liability for non-cash issuance costs |
| $ | — |
|
| $ |
| |
Seller financed portion of investment in PET Labs Pharmaceuticals |
| $ |
|
| $ | - |
| |
Purchase of property and equipment included in accounts payable |
| $ |
|
| $ |
| ||
Right-of-use assets obtained in exchange for lease liability |
| $ |
|
| $ | — |
| |
The accompanying notes are an integral part of these consolidated financial statements.
| 67 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements
1. Organization
Description of Business
ASP Isotopes Inc. was incorporated in the state of Delaware on September 13, 2021 and has its principal operations in Washington, DC. ASP Isotopes Inc.’s subsidiary, ASP Isotopes Holdings Limited (“ASP Guernsey”), has its principal operations in Guernsey. ASP Guernsey’s subsidiary, ASP Isotopes Holdings South Africa Proprietary Limited (“ASP South Africa”), has its principal operations in South Africa. ASP Isotopes UK Ltd, a wholly-owned subsidiary of the Company, was incorporated in July 2022. Enriched Energy, LLC, a wholly-owned subsidiary of the Company, was incorporated in January 2022. ASP Rentals Proprietary Limited (“ASP Rentals”) a variable interest entity (“VIE”) of ASP South Africa, has its principal operations in South Africa. ASP Isotopes Inc., its subsidiaries and ASP Rentals are collectively referred to as “the Company” throughout these consolidated statements.
The Company is a development stage advanced materials company dedicated to the development of technology and processes that, if successful, will allow for the enrichment of natural isotopes into higher concentration products, which could be used in several industries. The Company has an exclusive license to use proprietary technology, the Aerodynamic Separation Process (“ASP technology”), originally developed and licensed to the Company by Klydon Proprietary Ltd (“Klydon”), for the production, distribution, marketing and sale of all isotopes. The Company’s initial focus is on the production and commercialization of enriched Carbon-14 (“C-14”), Molybdenum-100 (“Mo-100”) and Silicon-28 (“Si-28”). Klydon has agreed to provide the Company a first commercial-scale isotope enrichment plant located in South Africa. The Company believes the C-14 it may develop using the ASP technology may be used in the development of new pharmaceuticals and agrochemicals. The Company believes that the Mo-100 it may develop using the ASP technology has significant potential advantages for use in the preparation of nuclear imaging agents by radiopharmacies and others in the medical industry. The Company believes the Si-28 it may develop using the ASP technology may be used to develop advanced semiconductors and in quantum computing.
The Company also intends to use the ASP technology to produce enriched Uranium-235 (“U-235”). The Company believes that the U-235 it may develop using the ASP technology may be commercialized as a nuclear fuel component for use in the new generation of HALEU-fueled small modular reactors that are now under development for commercial and government uses. In addition, the Company is considering future development of the ASP technology for the separation of Zinc-68, Ytterbium-176, Zinc-67, Nickel-64 and Xenon-136 for potential use in the healthcare target end market, and Chlorine -37 and Lithium-6 for potential use in the nuclear energy target end market.
In November 2022, the Company completed its IPO, selling an aggregate of
Liquidity and Going Concern Uncertainty
The accompanying consolidated financial statements have been prepared on a basis which assumes the Company is a going concern and do not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts and classifications of liabilities that may result from any uncertainty related to the Company’s ability to continue as a going concern. Such adjustments could be material. The Company has experienced net losses and negative cash flows from operating activities since its inception. The Company incurred net losses of $
The Company currently expects that its cash of $
There can be no assurance that the Company will achieve or sustain positive cash flows from operations or profitability. The Company is in the process of seeking additional debt and equity financing. However, such funding may not be available on a timely basis on terms acceptable to the Company, or at all. If the Company is unable to raise additional capital when required or on acceptable terms, the Company may be required to further scale back or discontinue the advancement of product candidates, further reduce headcount, reorganize, merge with another entity, or cease operations.
| 68 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
2. Basis of Presentation and Summary of Significant Accounting Policies
Basis of Presentation and Use of Estimates
The Company’s consolidated financial statements are prepared in accordance with accounting principles generally accepted in the United States (“GAAP”). The preparation of the Company’s consolidated financial statements requires management to make estimates and assumptions that impact the reported amounts of assets, liabilities and expenses and disclosure in the Company’s consolidated financial statements and accompanying notes. The most significant estimates in the Company’s consolidated financial statements relate to stock based compensation and the accounting for the acquisition. Although these estimates are based on the Company’s knowledge of current events and actions it may undertake in the future, actual results may materially differ from these estimates and assumptions.
Principles of Consolidation
The Company’s consolidated financial statements include the accounts of ASP Isotopes Inc., its subsidiaries and, beginning in 2023, the 80% owned Enlightened Isotopes, the 51% owned PET Labs Pharmaceuticals (see Note 11) and the 24% owned ASP Rentals (see Note 11). All intercompany balances and transactions have been eliminated in consolidation. For the year ended December 2023, there was no corporate activity for Enlightened Isotopes other than its formation and therefore there was no non-controlling interest to report on the consolidated balance sheet and no net loss attributable to non-controlling interest on the consolidated statement of operations and comprehensive loss to report. See Note 10.
Currency and Currency Translation
The consolidated financial statements are presented in U.S. dollars, the Company’s reporting currency. The functional currency of ASP Isotopes Inc. and ASP Guernsey is the U.S. dollar. The functional currency of the Company’s subsidiary ASP South Africa and PET Labs Pharmaceuticals is the South African Rand. Adjustments that arise from exchange rate changes on transactions of each group entity denominated in a currency other than the functional currency are included in other income and expense in the consolidated statements of operations. Assets and liabilities of ASP South Africa and PET Labs Pharmaceuticals are recorded in their South African Rand functional currency and translated into the U.S. dollar reporting currency of the Company at the exchange rate on the balance sheet date. Revenue, when recorded, and expenses of ASP South Africa and PET Labs Pharmaceuticals are recorded in their South African Rand functional currency and translated into the U.S. dollar reporting currency of the Company at the average exchange rate prevailing during the reporting period. Resulting translation adjustments are recorded to other comprehensive income (loss).
Concentration of Credit Risk and other Risks
Cash balances are maintained at U.S. financial institutions and may exceed the Federal Deposit Insurance Corporation (“FDIC”) insurance limit of $
Our foreign subsidiaries held cash of approximately $
Cash
The Company considers all highly liquid investments with original maturities at the date of purchase of three months or less to be cash equivalents. Cash and cash equivalents are stated at fair value and may include money market funds, U.S. Treasury and U.S. government-sponsored agency securities, corporate debt, commercial paper and certificates of deposit. The Company had no cash equivalents as of December 31, 2023 and 2022.
| 69 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
Segment Information
As of December 31, 2023, the Company manages its operations as a single segment for the purposes of assessing performance and making operating decisions, specialist isotopes and related services. The financial information is regularly reviewed by the chief operating decision maker (“CODM”) in deciding how to allocate resources. The Company’s CODM is its chief executive officer.
Fair Value of Financial Instruments
Accounting guidance defines fair value, establishes a consistent framework for measuring fair value, and expands disclosure for each major asset and liability category measured at fair value on either a recurring or nonrecurring basis. Fair value is defined as an exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants. As such, fair value is a market-based measurement that should be determined based on assumptions that market participants would use in pricing an asset or liability. As a basis for considering such assumptions, the accounting guidance establishes a three-tier fair value hierarchy, which prioritizes the inputs used in measuring fair value as follows:
| Level 1: | Observable inputs such as quoted prices in active markets; |
|
|
|
| Level 2: | Inputs, other than the quoted prices in active markets, that are observable either directly or indirectly; and |
|
|
|
| Level 3: | Unobservable inputs in which there is little or no market data, which require the reporting entity to develop its own assumptions. |
There was no share liability as of December 31, 2023. The Company’s share liability (Note 12) measured at Level 3 fair value on a recurring basis was $
|
| Share Liability |
| |
Balance as of December 31, 2021 |
| $ |
| |
Additional liability for issuance of common stock |
|
|
| |
Fair value adjustment |
|
| ( | ) |
Balance as of December 31, 2022 |
|
|
| |
Additional liability for issuance of common stock |
|
|
| |
Settlement of share liability with issuance of common stock |
|
| ( | ) |
Fair value adjustment |
|
|
| |
Balance as of December 31, 2023 |
| $ |
| |
The carrying amounts of accounts payable, accrued expenses and notes payable are considered to be representative of their respective fair values because of the short-term nature of those instruments.
Revenue Recognition
The Company’s revenue relates to PET Labs Pharmaceuticals, in which the Company acquired 51% ownership on October 31, 2023 (Note 11). The Company recognizes revenue in accordance with ASC Topic 606, Revenue from Contracts with Customers (“ASC 606”). The Company enters into transactions with radiopharmacy companies that are within the scope of ASC 606. The terms of these transactions include payment for delivery of nuclear medical doses for PET scanning in South Africa.
| 70 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
Under ASC 606, an entity recognizes revenue when its customer obtains control of promised goods or services, in an amount that reflects the consideration which the entity expects to receive in exchange for those goods or services. To determine the appropriate amount of revenue to be recognized for arrangements determined to be within the scope of ASC 606, the Company performs the following five steps: (i) identification of the promised goods or services in the contract; (ii) determination of whether the promised goods or services are performance obligations including whether they are distinct in the context of the contract; (iii) measurement of the transaction price, including the constraint on variable consideration; (iv) allocation of the transaction price to the performance obligations; and (v) recognition of revenue when (or as) the Company satisfies each performance obligation. The Company only applies the five-step model to contracts when it is probable that the entity will collect consideration it is entitled to in exchange for the goods or services it transfers to the customer.
The Company’s evaluates a transaction’s performance obligations to determine if promised goods or services in a contract to transfer a distinct good or service to the customer and are considered distinct when (i) the customer can benefit from the good or service on its own or together with other readily available resources and (ii) the promised good or service is separately identifiable from other promises in the contract. In assessing whether promised goods or services are distinct, the Company considers whether the goods or services are integral or dependent to other goods or services in the contract.
The Company determines the transaction price based on the agreed government rates for the promised goods in the contract.
The consideration is recognized as revenue when control is transferred for the related goods.
The Company evaluates the measure of progress each reporting period and, if necessary, adjusts the measure of performance and related revenue recognition. The Company receives payments from its customers based on billing schedules established in each contract. Upfront payments and fees are recorded as deferred revenue upon receipt or when due until the Company performs its obligations under these arrangements. Amounts are recorded as accounts receivable when the Company’s right to consideration is unconditional.
Accounts Receivable
Accounts receivable are stated at the amount management expects to collect from outstanding balances. An allowance for expected credit losses is estimated for those accounts receivable considered to be uncollectible based upon historical experience and management's evaluation of outstanding accounts receivable. We maintain an allowance for expected credit losses for accounts receivable, which is recorded as an offset to accounts receivable, and changes in such are classified as selling, general and administrative expense in the Consolidated Statements of Operations and Comprehensive Loss. We assess collectibility by reviewing accounts receivable on a collective basis where similar characteristics exist and on an individual basis when we identify specific customers with known disputes or collectibility issues. In determining the amount of the allowance for credit losses, we consider historical collectibility based on past due status and make judgments about the creditworthiness of customers based on ongoing credit evaluations. We also consider customer-specific information, current market conditions, and reasonable and supportable forecasts of future economic conditions. Bad debts are written off against the allowance when identified. At December 31, 2023 and 2022 there was no allowance for expected credit losses.
| 71 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
Property and Equipment
Property and equipment include costs of assets constructed, purchased or leased under a finance lease, related delivery and installation costs and interest incurred on significant capital projects during their construction periods. Expenditures for renewals and betterments also are capitalized, but expenditures for normal repairs and maintenance are expensed as incurred. Costs associated with yearly planned major maintenance are generally deferred and amortized over 12 months or until the same major maintenance activities must be repeated, whichever is shorter. The cost and accumulated depreciation applicable to assets retired or sold are removed from the respective accounts, and gains or losses thereon are included in the statement of operations.
We assign the useful lives of our property and equipment based upon our internal engineering estimates, which are reviewed periodically. The estimated useful lives of our property and equipment range from
Construction in progress (see Note 4) is carried at cost and consists of specifically identifiable direct and indirect development and construction costs. While under construction, costs of the property are included in construction in progress until the property is placed in service, at which time costs are transferred to the appropriate property and equipment account, including, but not limited to, leasehold improvements or other such accounts.
Property and equipment acquired from the PET Labs Pharmaceutical Acquisition was measured at fair value on October 31, 2023. The fair value forms the new basis of these assets and is depreciated over the remaining estimated useful lives of the related assets.
Business Combination and Asset Acquisitions
The Company evaluates acquisitions of assets and other similar transactions to assess whether or not the transaction should be accounted for as a business combination or asset acquisition by first applying a screen to determine if substantially all of the fair value of the gross assets acquired is concentrated in a single identifiable asset or group of similar identifiable assets. If the screen is met, the transaction is accounted for as an asset acquisition. If the screen is not met, further determination is required as to whether or not the Company has acquired inputs and processes that have the ability to create outputs, which would meet the requirements of a business. If determined to be a business combination, the Company accounts for the transaction under the acquisition method of accounting in accordance with ASC 805 Business Combinations, which requires the acquiring entity in a business combination to recognize the fair value of all assets acquired, liabilities assumed, and any non-controlling interest in the acquiree and establishes the acquisition date as the fair value measurement point. Accordingly, the Company recognizes assets acquired and liabilities assumed in business combinations, including contingent assets and liabilities, and non-controlling interest in the acquiree based on the fair value estimates as of the date of acquisition. In accordance with ASC 805, the Company recognizes and measures goodwill as of the acquisition date, as the excess of the fair value of the consideration paid over the fair value of the identified net assets acquired.
The consideration for the Company’s business acquisitions may include future payments that are contingent upon the occurrence of a particular event or events. The obligations for such contingent consideration payments are recorded at fair value on the acquisition date. The contingent consideration obligations are then evaluated each reporting period. Changes in the fair value of contingent consideration, other than changes due to payments, are recognized as a gain or loss and recorded within change in the fair value of deferred and contingent consideration liabilities in the consolidated statements of comprehensive loss.
If determined to be an asset acquisition, the Company accounts for the transaction under ASC 805-50, which requires the acquiring entity in an asset acquisition to recognize assets acquired and liabilities assumed based on the cost to the acquiring entity on a relative fair value basis, which includes transaction costs in addition to consideration given. No gain or loss is recognized as of the date of acquisition unless the fair value of non-cash assets given as consideration differs from the assets’ carrying amounts on the acquiring entity’s books. Consideration transferred that is non-cash will be measured based on either the cost (which shall be measured based on the fair value of the consideration given) or the fair value of the assets acquired and liabilities assumed, whichever is more reliably measurable. Goodwill is not recognized in an asset acquisition and any excess consideration transferred over the fair value of the net assets acquired is allocated to the identifiable assets based on relative fair values.
Contingent consideration payments in asset acquisitions are recognized when the contingency is resolved and the consideration is paid or becomes payable (unless the contingent consideration meets the definition of a derivative, in which case the amount becomes part of the basis in the asset acquired). Upon recognition of the contingent consideration payment, the amount is included in the cost of the acquired asset or group of assets.
| 72 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
Goodwill
Goodwill represents the amount of consideration paid in excess of the fair value of net assets acquired as a result of the Company’s business acquisitions accounted for using the acquisition method of accounting. Goodwill is not amortized and is subject to impairment testing at a reporting unit level on an annual basis or when a triggering event occurs that may indicate the carrying value of the goodwill is impaired. An entity is permitted to first assess qualitative factors to determine if a quantitative impairment test is necessary. Further testing is only required if the entity determines, based on the qualitative assessment, that it is more likely than not that the fair value of the reporting unit is less than its carrying amount. The Company will perform its annual test for goodwill as of October 31.
Leases
The Company accounts for leases in accordance with ASC 842, Leases. At the inception of an arrangement, the Company determines whether the arrangement is or contains a lease based on specific facts and circumstances, the existence of an identified asset(s), if any, and the Company’s control over the use of the identified asset(s), if applicable. Operating lease liabilities and their corresponding right-of-use assets are recorded based on the present value of future lease payments over the expected lease term. The interest rate implicit in lease contracts is typically not readily determinable. As such, the Company will utilize the incremental borrowing rate, which is the rate incurred to borrow on a collateralized basis over a similar term an amount equal to the lease payments in a similar economic environment, and considering the region in which the ROU asset and liabilities are located.
The Company has elected to combine lease and non-lease components as a single component. Operating leases are recognized on the balance sheet as ROU lease assets, operating lease liabilities current and operating lease liabilities non-current. Fixed rents are included in the calculation of the lease balances, while variable costs paid for certain operating and pass-through costs are excluded. Lease expense is recognized over the expected term on a straight-line basis.
Finance leases are recognized on the balance sheet as property and equipment, finance lease liabilities current and finance lease liabilities non-current. Finance lease ROU assets and the related lease liabilities are recognized based on the present value of the future minimum lease payments over the lease term at commencement date. The finance lease ROU assets are amortized on a straight-line basis over the lease term with the related interest expense of the lease liability payment recognized over the lease term using the effective interest method.
Impairment of Long-lived Assets
Long-lived assets consist primarily of property and equipment. The Company reviews long-lived assets for impairment whenever events or changes in circumstances indicate the carrying amount of an asset is not recoverable. Recoverability of assets to be held and used is measured by a comparison of the carrying amount of an asset to the future undiscounted net cash flows expected to be generated by the asset. If such assets are considered to be impaired, the impairment to be recognized is measured as the amount by which the carrying amount of the asset exceeds the fair value of the assets. Fair value would be assessed using a discounted cash flows or other appropriate measures of fair value. The Company did not recognize any impairment losses for the years ended December 31, 2023 and 2022.
Research and Development Costs
Research and development costs consist primarily of fees paid to consultants, license fees and facilities costs. Nonrefundable advance payments for goods and services that will be used in future research and development activities are expensed when the activity has been performed or when the goods have been received rather than when the payment is made. All research and development costs are expensed as incurred.
Selling, General and Administrative Costs
Selling, general and administrative expenses consist primarily of salaries and related benefits, including stock-based compensation, related to our executive, finance, business development, legal, human resources and support functions. Other general and administrative expenses include professional fees for auditing, tax, consulting and patent-related services, rent and utilities and insurance.
| 73 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
Stock-based Compensation
Stock-based compensation expense represents the cost of the grant date fair value of employee stock awards recognized over the requisite service period of the awards (usually the vesting period) on a straight-line basis. The Company estimates the fair value of each stock-based award on the date of grant using the Black-Scholes option pricing model. The Black-Scholes option pricing model incorporates various assumptions, such as the value of the underlying common stock, the risk-free interest rate, expected volatility, expected dividend yield, and expected life of the options. Forfeitures are recognized as a reduction of stock-based compensation expense as they occur.
The Company also awards restricted stock to employees and directors. Restricted stock is generally subject to forfeiture if employment terminates prior to the completion of the vesting restrictions. The Company expenses the cost of the restricted stock, which is determined to be the fair market value of the shares of common stock underlying the restricted stock at the date of grant, ratably over the period during which the vesting restrictions lapse.
Equity-based compensation expense is classified in the statement of operations in the same manner in which the award recipients’ payroll costs are classified or in which the award recipients’ service payments are classified.
Prior to the Company’s IPO, there was no public market of the Company’s common stock. The fair value of the shares of common stock underlying the Company’s share-based awards was estimated on each grant date by the Company’s board of directors based on then current facts and circumstances. To determine the fair value of the Company’s common stock underlying option grants, the board of directors considered, among other things, input from management and recent third-party financings consummated by the Company.
Income Taxes
Deferred income tax assets and liabilities arise from temporary differences associated with differences between the financial statements and tax basis of assets and liabilities, as measured by the enacted tax rates, which are expected to be in effect when these differences reverse. Valuation allowances are established when necessary to reduce deferred tax assets to the amount expected to be realized.
Prior to the acquisition of 51% of PET Labs Pharmaceuticals, the Company had generated net losses since inception and accordingly had not recorded a provision for income taxes. Subsequent to the acquisition of 51% of PET Labs Pharmaceuticals, the Company records the provision for income taxes for the activity from PET Labs Pharmaceuticals operations.
The Company follows the provisions of ASC 740-10, Uncertainty in Income Taxes, or ASC 740-10. The Company has not recognized a liability for any uncertain tax positions. A reconciliation of the beginning and ending amount of unrecognized tax benefits has not been provided since there is no unrecognized benefit since the date of adoption. The Company has not recognized interest expense or penalties as a result of the implementation of ASC 740-10. If there were an unrecognized tax benefit, the Company would recognize interest accrued related to unrecognized tax benefits and penalties in income tax expense.
The Company has identified the United States, South Africa and Guernsey as its major tax jurisdictions. Refer to Note 15 for further details.
Comprehensive Loss
Comprehensive loss is defined as a change in equity during a period from transactions and other events and circumstances from non-owner sources. The Company’s comprehensive loss is comprised of net loss and the effect of currency translation adjustments.
Recently Issued Accounting Pronouncements
The Company has reviewed recently issued accounting pronouncements and plans to adopt those that are applicable to it. The Company does not expect the adoption of any recently issued pronouncements to have a material impact on its results of operations or financial position.
| 74 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
3. Revenue
In connection with our acquisition of
The following table presents changes in the Company’s accounts receivable from the PET Labs Pharmaceuticals acquisition date of October 31, 2023 through December 31, 2023:
|
| Balance as of October 31, 2023 |
|
| Additions |
|
| Deductions |
|
| Balance as of December 31, 2023 |
| ||||
|
|
|
|
|
|
|
|
|
|
|
|
| ||||
Accounts receivable |
| $ |
|
| $ |
|
| $ | ( | ) |
| $ |
| |||
4. Property and Equipment
Property and equipment as of December 31, 2023 and 2022 consisted of the following:
| Useful Lives (Years) |
|
| December 31, |
| ||||||
|
|
|
|
| 2023 |
|
| 2022 |
| ||
Construction in progress |
| - |
|
| $ |
|
| $ |
| ||
Tools, machinery and equipment |
|
|
|
|
|
|
|
| |||
Computer equipment |
|
|
|
|
|
|
|
| |||
Vehicles |
| |
|
|
|
|
|
|
| ||
Software |
| |
|
|
|
|
|
|
| ||
Office furniture |
| |
|
|
|
|
|
|
| ||
Leasehold improvements |
| |
|
|
|
|
|
|
| ||
Property and equipment, at cost |
|
|
|
|
|
|
|
|
| ||
Less accumulated depreciation |
|
|
|
|
| ( | ) |
|
|
| |
Property and equipment, net |
|
|
|
| $ |
|
| $ |
| ||
The Company is currently building out plants in Pretoria, South Africa and all costs incurred are considered construction in progress because the work is not complete as of December 31, 2023 and 2022. There was no depreciation expense as it relates to the construction in progress for the year ended December 31, 2023 and 2022. Depreciation expense for all other asset categories was $
5. Accrued Expenses
Accrued expenses as of December 31, 2023 and 2022 consisted of the following:
|
| December 31, |
| |||||
|
| 2023 |
|
| 2022 |
| ||
Accrued professional |
| $ |
|
| $ |
| ||
Accrued salaries and other employee costs |
|
|
|
|
|
| ||
Accrued other |
|
|
|
|
|
| ||
Total accrued expenses |
| $ |
|
| $ |
| ||
| 75 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
6. Notes Payable
During 2021, the Company executed promissory notes payable with two individuals with an aggregate principal balance of approximately $
In March 2022, one of the promissory notes totaling $
In conjunction with the acquisition of
In November 2023, the Company executed a promissory note payable with a finance company for $
7. Deferred Revenues
In June 2023, the Company entered into a Supply Agreement with a customer for the delivery of molybdenum-100 and molybdenum-98 beginning in 2024. In conjunction with the Supply Agreement, the Company received $
8. Commitments and Contingencies
Purchase of Cyclotron
In November 2023, the cyclotron that the Company ordered was shipped. As of December 31, 2023 the equipment had not been delivered. The Company is obligated to purchase this equipment and recorded the other asset and other liability for the full cost of $
Klydon Proprietary Limited
In November 2021, the Company entered into an agreement with Klydon Proprietary Limited (“Klydon”) to design and build a plant to enrich Molybdenum in South Africa. The initial phase of the project includes the building of a plant that can support the production of at least 5kgs of Mo-100, and is expected to be completed in the second half of 2023. The contracted cost for this phase is $
Klydon performed a portion of the services required under the Turnkey Contract; however, services were incomplete and many of the services were not completed within the time frame required. As a result, Klydon and ASP South Africa entered into an Acknowledgement of Debt Agreement dated November 30, 2022, whereby Klydon (i) agreed to pledge its assets (the “Pledged Assets”) to ASP South Africa to secure its performance of the Turnkey Contract by December 31, 2022, and (ii) acknowledged that ASP South Africa would suffer damages in the amount of $
On April 4, 2023, the Company perfected its interest under the Acknowledgement of Debt Agreement, pursuant to which the Company acquired certain intellectual property from Klydon (“Klydon Settlement”). In addition, the Company acquired Klydon’s interest in four entities which are inactive and in the process of being dissolved. The Company has concluded that the Klydon Settlement is accounted for under ASC 805, Business Combinations as an asset acquisition since the assets acquired were concentrated in a single identifiable asset from a related party. In conjunction with the Klydon Settlement, the Company recorded an increase to additional paid-in capital for the settlement of all liabilities owed to Klydon at the time of settlement totaling $
Two individuals who are officers and board members of Klydon, one who is now an officer of ASP Isotopes Inc. and the other who is now a scientific advisor of ASP Isotopes Inc., received warrants to purchase common stock of the Company and therefore are considered related parties. See Notes 10 and 12.
| 76 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
Contingencies
From time to time, the Company may have certain contingent liabilities that arise in the ordinary course of its business activities. The Company accrues liabilities for such matters when future expenditures are probable and such expenditures can be reasonably estimated.
On October 25, 2022, the Company received a letter from a law firm acting on behalf of Norsk medisinsk syklotronsenter AS (“NMS”), asserting, among other things, that the grant of a license to the ASP technology to the Company by Klydon violates a pre-existing exclusive sub-license to the ASP technology granted to Radfarma. The asserted claims, arbitration and/or litigation could include claims against the Company, the Company’s licensor (Klydon), or Klydon’s present or former sub-licensors alleging infringement of intellectual property rights with respect to the ASP technology on which our company relies. The Company recorded legal costs totaling $
9. Leases
The Company accounts for facility leases in accordance with ASC 842 (Note 2). The Company is party to five facility leases in South Africa for office, manufacturing and laboratory space.
A lease for office and laboratory space in Pretoria, South Africa commenced in October 2021 with the initial term set to expire in
A lease for additional production space in Pretoria, South Africa commenced in April 2023 with the initial term expiring in March 2024 and the Company is maintaining the lease under the agreed upon monthly extensions. The Company has applied the guidance in ASC 842 and has determined that this lease is a short term lease and expenses the monthly payments as incurred.
A lease for laboratory space in Pretoria, South Africa commenced in November 2023 with the initial term set to expire in
A lease for office and production space in Pretoria, South Africa commenced prior to October 31, 2023 with the initial term set to expire in
A lease for additional production space in Pretoria, South Africa commenced prior to October 31, 2023 with the initial term expiring in March 2024 and the Company is maintaining the lease under the agreed upon monthly extensions. The Company has applied the guidance in ASC 842 and has determined that this lease is a short term lease effective on the date of ASP Isotopes acquisition of 51% of PET Labs Pharmaceuticals and expensed the monthly payments for the two months ended December 31, 2023.
| 77 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
Quantitative information regarding the Company’s operating lease liabilities is as follows:
|
| Year Ended December 31, 2023 |
|
| Year Ended December 31, 2022 |
| ||
Operating Lease Cost |
|
|
|
|
|
| ||
Operating lease cost |
| $ |
|
| $ |
| ||
Other Information |
|
|
|
|
|
|
|
|
Operating cash flows paid for amounts included in the measurement of lease liabilities |
| $ |
|
| $ |
| ||
Operating lease liabilities arising from obtaining right-of-use assets |
| $ |
|
| $ |
| ||
Weighted average remaining lease term (years) |
|
|
|
|
| |||
Weighted average discount rate |
|
| % |
|
| % | ||
Future lease payments under noncancelable operating lease liabilities are as follows as of December 31, 2023:
|
| Operating Leases |
| |
Future Lease Payments |
|
|
| |
2024 |
| $ |
| |
2025 |
|
|
| |
2026 |
|
|
| |
2027 |
|
|
| |
2028 |
|
|
| |
Thereafter |
|
|
| |
Total lease payments |
| $ |
| |
Less: imputed interest |
|
| ( | ) |
Total lease liabilities |
| $ |
| |
Less current portion |
|
| ( | ) |
Lease liability – noncurrent |
| $ |
| |
The Company records the expense from short term leases as incurred. For the year ending December 31, 2023, the Company recorded $
The Company accounts for finance leases in accordance with ASC 842 (Note 2). Subsequent to the acquisition of 51% of PET Labs Pharmaceuticals, the Company is party to nine finance leases in South Africa for certain fixed assets.
Quantitative information regarding the Company’s finance lease liabilities is as follows:
|
| Year Ended December 31, 2023 |
|
| Year Ended December 31, 2022 |
| ||
Finance Lease Cost |
|
|
|
|
|
| ||
Interest on lease liabilities |
| $ |
|
| $ |
| ||
Other Information |
|
|
|
|
|
|
|
|
Operating cash flows paid for amounts included in the measurement of finance lease liabilities |
| $ |
|
| $ |
| ||
Amortization of right-of-use assets |
| $ |
|
| $ |
| ||
Weighted average remaining lease term (years) |
|
|
|
|
| — |
| |
Weighted average discount rate |
|
| % |
| % | |||
Future lease payments under noncancelable finance lease liabilities are as follows as of December 31, 2023:
|
| Finance Leases |
| |
Future Lease Payments |
|
|
| |
2024 |
| $ |
| |
2025 |
|
|
| |
2026 |
|
|
| |
2027 |
|
|
| |
2028 |
|
|
| |
Thereafter |
|
|
| |
Total lease payments |
| $ |
| |
Less: imputed interest |
|
| ( | ) |
Total lease liabilities |
| $ |
| |
Less current portion |
|
| ( | ) |
Lease liability – noncurrent |
| $ |
| |
| 78 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
10. License Agreements
In September 2021, the Company licensed certain intellectual property from Klydon for the development, production distribution, marketing and sale of Mo-100. The license term is 999 years, unless terminated earlier by either party under certain provisions. Any development efforts improving the intellectual property performed by either Klydon or the Company will be the property of Klydon. There are no upfront, milestone payments, nor royalties on product sales over the term of the license. Two individuals who are officers and board members of Klydon received warrants to purchase common stock of the Company. (See Note 12.)
In January 2022, the Company licensed certain intellectual property from Klydon for the development, production distribution, marketing and sale of uranium isotope U-235 (“U-235”). The license term is 999 years, unless terminated earlier by either party under certain provisions. Any development efforts improving the intellectual property performed by either Klydon or the Company will be the property of Klydon. The Company paid an upfront fee of $
In July 2022, ASP Isotopes UK Ltd (a subsidiary of the Company) entered into a license agreement with Klydon, as licensor, pursuant to which ASP Isotopes UK Ltd acquired from Klydon an exclusive license to use, develop, modify, improve, subcontract and sublicense certain intellectual property rights relating to the ASP technology for the production, distribution, marketing and sale of all isotopes produced using the ASP technology (the “Klydon license agreement”). The Klydon license agreement superseded and replaced the Mo-100 license and U-235 license described in Note 8 above. The Klydon license agreement is royalty-free, has a term of 999 years and is worldwide for the development of the ASP technology and the distribution, marketing and sale of isotopes. Future production of isotopes is limited to member countries of the Nuclear Suppliers Group. In connection with the Klydon license agreement the Company agreed to make an upfront payment of $
In July 2022, ASP South Africa acquired assets comprising a dormant Silicon-28 aerodynamic separation processing plant from Klydon for ZAR
On April 4, 2023, the Company perfected its interest under the Acknowledgement of Debt Agreement (see Note 8), pursuant to which the Company acquired certain intellectual property from Klydon (“Klydon Settlement”). The Company concluded that the Klydon Acquisition is accounted for under ASC 805, Business Combinations as an asset acquisition since the assets acquired were concentrated in a single identifiable asset from a related party. In conjunction with the Klydon Settlement, the Company recorded an increase to additional paid-in capital for the settlement of all liabilities owed to Klydon at the time of settlement totaling $
11. Acquisitions
PET Labs Pharmaceuticals
In October 2023, the Company completed the PET Labs Pharmaceuticals Acquisition, a provider of nuclear medical doses for use in PET scans in South Africa. The acquisition of PET Labs Pharmaceuticals was intended to accelerate the distribution of the Company’s pipeline. The acquisition of PET Labs Pharmaceuticals has been accounted for as a business combination in accordance with ASC 805.
Pursuant to the terms of the agreement, the Company acquired
In addition to the purchase consideration, the Company has an option to purchase the remaining
| 79 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
The Company incurred approximately $
Dr. Gerdus Kemp is an officer of PET Labs Pharmaceuticals and, effective November 1, 2023, an employee of ASP Isotopes UK Ltd. In addition, Dr. Kemp controls the remaining 49% ownership of PET Labs Pharmaceuticals.
The following table summarizes the preliminary allocation of the purchase consideration to the fair value of the assets acquired and liabilities assumed:
Consideration
Cash |
| $ |
| |
Present value of balance due |
|
|
| |
|
| $ |
|
Recognized amounts of identifiable assets acquired and liabilities assumed
Cash and cash equivalents |
| $ |
| |
Accounts receivable |
|
|
| |
Other current assets |
|
|
| |
Property and equipment |
|
|
| |
Right of use assets |
|
|
| |
Financial liabilities |
|
| ( | ) |
Right of use liabilities |
|
| ( | ) |
Total identifiable net assets |
|
|
|
Noncontrolling interest |
|
| (1,821,021 | ) |
Goodwill |
|
|
| |
|
| $ |
|
Goodwill arising from the acquisition as of October 31, 2023 of $
The Company considered the contractual value of accounts receivable to be the same as the fair value and expects the full amount to be collected.
The results of PET Labs Pharmaceuticals have been included in the consolidated financial statements from the date of the acquisition.
The changes to the carrying value of goodwill is as follows:
Balance as of October 31, 2023 (acquisition date) |
| $ |
| |
Translation adjustment |
|
| |
|
Balance as of December 31, 2023 |
| $ |
|
Unaudited Pro Forma Financial Information
The following unaudited pro forma financial information shows the results of the Company’s operations for the years ended December 31, 2023 and 2022 as if the acquisition had occurred on January 1, 2022. The unaudited pro forma financial information is presented for information purposes only and is not necessarily indicative of the Company’s performance had the acquisition occurred as of that date. The unaudited pro forma information is also not intended to be a projection of future results due to the integration of the acquired operations of PET Labs Pharmaceuticals. The unaudited pro forma information reflects the effects of applying the Company’s accounting policies to the combined historical financial information of the Company and PET Labs Pharmaceuticals.
|
| Pro Forma |
| |||||
|
| 2023 |
|
| 2022 |
| ||
|
|
|
|
| ||||
Revenue |
| $ |
|
| $ |
| ||
Net loss |
| $ | ( | ) |
| $ | ( | ) |
Net loss per common share |
| $ | ( | ) |
| $ | ( | ) |
| 80 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
ASP Rentals
In December 2023, the Company entered into Shareholders Agreement (“ASP Rentals Shareholders Agreement”) with ASP Rentals, an equipment financing service provider in South Africa. In conjunction with the ASP Rental Shareholders Agreement, the Company entered into an Asset Sale Agreement and an Asset Rental Agreement in order to facilitate the financing of energy equipment recently purchased by ASP South Africa. ASP Rentals is considered a variable interest entity, and the Company is the primary beneficiary and therefore ASP Rentals has been consolidated in accordance with ASC 810.
As of December 31, 2023, ASP Rentals has a receivable and an obligation to issue
Consideration for all common shares of ASP Rentals was received in January 2024.
12. Stockholders’ Equity
Preferred stock
The Company has
Common stock
The Company has
During 2022, prior to the IPO, the Company issued
In November 2022, the Company was required to issue shares of common stock with a then fair value totaling $
In November 2022, the Company completed its IPO, selling an aggregate of
| 81 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
In February 2023, the Company was required to issue an aggregate of
In March 2023, the Company was required to issue an aggregate of
In March 2023, an officer and scientific advisor of the Company exchanged an aggregate of
In March 2023, the Company’s non-employee board members agreed to receive the 2022 cash director fees totaling $
In March 2023, the Company issued
In May 2023, the Company was required to issue an aggregate of
In May 2023, the Company was required to issue an aggregate of
In July 2023, the Company was required to issue an aggregate of
In August 2023, the Company was required to issue an aggregate of
In October 2023, the Company entered into Securities Purchase Agreements with certain institutional and other accredited investors and certain directors of the Company to issue and sell an aggregate of
Activity of the share liabilities for the year ended December 31, 2023 is as follows:
|
| Share Liability as of December 31, 2022 |
|
| New Share Liabilities in 2023 |
|
| Mark to Market Adjustments in 2023 |
|
| Liabilities Settled in 2023 |
|
| Share Liabilities as of December 31, 2023 |
| |||||
Share liabilities originated in 2022 |
| $ |
|
| $ |
|
| $ | ( | ) |
| $ | ( | ) |
| $ |
| |||
Share liabilities originated in 2023 |
|
|
|
|
|
|
|
|
|
|
|
| ( | ) |
| $ |
| |||
|
| $ |
|
| $ |
|
|
|
|
|
| ( | ) |
| $ |
| ||||
| 82 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
Common Stock Warrants
In September 2021, the Company issued warrants to purchase
The fair values of the warrants to purchase
Expected volatility |
|
| % | |
Weighted-average risk-free rate |
|
| % | |
Expected term in years |
|
|
| |
Expected dividend yield |
|
| % |
13. Stock Compensation Plan
Equity Incentive Plan
In October 2021, the Company adopted the 2021 Stock Incentive Plan (“2021 Plan”) that provided for the issuance of common stock to employees, nonemployee directors, and consultants. Recipients of incentive stock options are eligible to purchase shares of common stock at an exercise price equal to no less than the estimated fair market value of such stock on the date of grant. The 2021 Plan provided for the grant of incentive stock options, non-statutory stock options, restricted stock, restricted stock units, stock awards and stock appreciation rights. The maximum contractual term of options granted under the 2021 Plan is ten years. The maximum number of shares initially available for issuance under the 2021 Plan was
In November 2022, the Company adopted the 2022 Equity Incentive Plan (“2022 Plan”) that provides for the issuance of common stock to employees, nonemployee directors, and consultants. Recipients of incentive stock options are eligible to purchase shares of common stock at an exercise price equal to no less than the estimated fair market value of such stock on the date of grant. The 2022 Plan provides for the grant of incentive stock options, non-statutory stock options, restricted stock, restricted stock units, stock awards and stock appreciation rights. The maximum contractual term of options granted under the 2022 Plan is ten years. The number of shares of the Company’s common stock reserved for issuance under the 2022 Plan is equal to
Stock Options
The following table sets forth the activity for the Company’s stock options during the periods presented:
|
| Number of Options |
|
| Weighted- Average Exercise Price per Share |
|
| Weighted Average Remaining Contractual Term (in Years) |
|
| Aggregate Intrinsic Value |
| ||||
Outstanding as of December 31, 2021 |
|
|
|
| $ |
|
|
|
|
| $ |
| ||||
Granted |
|
|
|
| $ |
|
|
|
|
|
|
|
|
| ||
Forfeited |
|
| ( | ) |
| $ |
|
|
|
|
|
|
|
|
| |
Outstanding as of December 31, 2022 |
|
|
|
| $ |
|
|
|
|
| $ |
| ||||
Granted |
|
| - |
|
| $ |
|
|
|
|
|
|
|
|
| |
Forfeited |
|
| ( | ) |
| $ |
|
|
|
|
|
|
|
|
| |
Outstanding as of December 31, 2023 |
|
|
|
| $ |
|
|
|
|
| $ |
| ||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Exercisable as of December 31, 2023 |
|
|
|
| $ |
|
|
|
|
| $ |
| ||||
Vested or expected to vest as of December 31, 2023 |
|
|
|
| $ |
|
|
|
|
| $ |
| ||||
| 83 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
The fair values of the options granted were estimated based on the Black-Scholes model, using the following assumptions:
|
|
| Year Ended December 31, 2022 |
|
Expected volatility |
|
|
| |
Risk-free interest rate |
|
|
| |
Expected term in years |
|
|
| |
Expected dividend yield |
|
|
|
For the year ended December 31, 2022, the Company granted
The Company recorded stock compensation from options of $
Stock Awards
In October 2021, the Company issued
In October 2021, the Company issued
In July 2022, the Company issued
In July 2022, the Company issued
In November 2022, the Company issued
In December 2022, the Company issued an aggregate of
In March 2023, the Company issued an aggregate of
In August 2023, the Company issued
In October 2023, the Company was obligated to issue $
| 84 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
The Company recorded stock compensation from stock awards totaling $
The following table summarizes awards and vesting of restricted common stock:
|
| Number of Shares |
|
| Weighted Average Grant Date Fair Value Per Share |
| ||
Unvested as of December 31, 2021 |
|
|
|
| $ |
| ||
Granted |
|
|
|
| $ |
| ||
Vested |
|
| ( | ) |
| $ |
| |
Unvested as of December 31, 2022 |
|
|
|
| $ |
| ||
Granted |
|
|
|
| $ |
| ||
Vested |
|
| ( | ) |
| $ |
| |
Unvested as of December 31 2023 |
|
|
|
| $ |
| ||
Stock-based Compensation Expense
Stock-based compensation expense for all stock awards recognized in the accompanying consolidated statements of operations is as follows:
|
| Year Ended December 31, 2023 |
|
| Year Ended December 31, 2022 |
| ||
Selling, general and administrative |
| $ |
|
| $ |
| ||
Research and development |
|
|
|
|
|
| ||
Total |
| $ |
|
| $ |
| ||
14. Net Loss Per Share
The Company has reported losses since inception and has computed basic net loss per share attributable to common stockholders by dividing net loss attributable to common stockholders by the weighted-average number of shares of Common Stock outstanding for the period, without consideration for potentially dilutive securities. The Company computes diluted net loss per share of Common Stock after giving consideration to all potentially dilutive shares of common stock, including options to purchase common stock and warrants to purchase common stock, outstanding during the period determined using the treasury-stock and if-converted methods, except where the effect of including such securities would be antidilutive. Because the Company has reported net losses since inception, these potential shares of Common Stock have been anti-dilutive and basic and diluted loss per share were the same for all periods presented.
The following table sets forth the computation of basic and diluted net loss per share:
|
| Year Ended December 31, 2023 |
|
| Year Ended December 31, 2022 |
| ||
Numerator: |
|
|
|
|
|
| ||
Net loss attributable to ASP Isotopes shareholders |
| $ | ( | ) |
| $ | ( | ) |
Denominator: |
|
|
|
|
|
|
|
|
Weighted average common stock outstanding, basic and diluted |
|
|
|
|
|
| ||
Net loss per share, basic and diluted |
| $ | ( | ) |
| $ | ( | ) |
| 85 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
The following table sets forth the potentially dilutive securities that have been excluded from the calculation of diluted net loss per share because to include them would be anti-dilutive:
|
| Year Ended December 31, 2023 |
|
| Year Ended December 31, 2022 |
| ||
Options to purchase common stock |
|
|
|
|
|
| ||
Restricted stock |
|
|
|
|
|
| ||
Warrants to purchase common stock |
|
|
|
|
|
| ||
Total shares of common stock equivalents |
|
|
|
|
|
| ||
15. Income Taxes
The components of net loss before taxes are as follows:
|
| Year Ended December 31, 2023 |
|
| Year Ended December 31, 2022 |
| ||
Domestic |
| $ | ( | ) |
| $ | ( | ) |
Foreign |
|
| ( | ) |
|
| ( | ) |
Total net loss before taxes |
| $ | ( | ) |
| $ | ( | ) |
The effective tax rate of the Company’s provision for income taxes differs from the federal statutory rate for the year ended December 31, 2023 and 2022 as follows:
|
| Year Ended December 31, 2023 |
|
| Year Ended December 31, 2022 |
| ||
Tax computed at federal statutory rate |
|
| % |
|
| % | ||
Earnings in jurisdictions taxed at rates different from the statutory U.S. federal tax rate |
|
| ( | )% |
|
| ( | )% |
Non-deductible stock compensation expense |
|
| ( | )% |
| % | ||
Permanent differences |
|
| % |
|
| % | ||
Other |
|
| ( | )% |
|
| % | |
Valuation allowance |
|
| ( | )% |
|
| ( | )% |
Income tax expense |
|
| % |
|
| — |
| |
Deferred income taxes reflect the net tax effects of (a) temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes, and (b) operating losses and tax credit carryforwards. Significant components of deferred tax assets (liabilities) are as follows:
|
| December 31, |
| |||||
|
| 2023 |
|
| 2022 |
| ||
Deferred tax assets: |
|
|
|
|
|
| ||
Net operating loss carryforwards |
| $ |
|
| $ |
| ||
Capitalized R&D costs |
|
|
|
|
|
| ||
Share-based compensation |
|
|
|
|
|
| ||
Accruals and reserves |
|
|
|
|
|
| ||
Right-of-use lease liability |
|
|
|
|
|
| ||
Total deferred tax assets |
|
|
|
|
|
| ||
|
|
|
|
|
|
|
|
|
Deferred tax liabilities: |
|
|
|
|
|
|
|
|
Property and equipment, net |
|
| ( | ) |
|
|
| |
Right-of-use lease asset |
|
| ( | ) |
|
| ( | ) |
Total deferred tax liabilities |
|
| ( | ) |
|
| ( | ) |
Total net deferred tax assets |
|
|
|
|
|
| ||
Less: valuation allowance |
|
| ( | ) |
|
| ( | ) |
Net deferred taxes (liabilities) assets |
| $ | ( | ) |
| $ |
| |
| 86 |
| Table of Contents |
ASP Isotopes Inc.
Notes to Consolidated Financial Statements (continued)
The Company recorded income tax income totaling $
As of December 31, 2023, the Company has federal, state and South Africa NOLs available of $
The Company recognizes a tax benefit from an uncertain tax position when it is more likely than not that the position will be sustained upon examination, including resolutions of any related appeals or litigation processes, based on the technical merits. Income tax positions must meet a more likely than not recognition threshold to be recognized. The Company’s practice is to recognize interest and/or penalties related to income tax matters in income tax expense. The Company had no accrual for interest and penalties on the Company’s balance sheets and has not recognized interest and/or penalties in the statements of operations and comprehensive loss for the years ended December 31, 2023 and 2022. Uncertain tax positions are evaluated based upon the facts and circumstances that exist at each reporting period. Subsequent changes in judgment based upon new information may lead to changes in recognition, derecognition, and measurement. Adjustments may result, for example, upon resolution of an issue with the taxing authorities or expiration of a statute of limitations barring an assessment for an issue. As of December 31, 2023 and December 31, 2022, there were no uncertain tax positions.
Ownership changes, as defined in the IRC, may limit the amount of net operating loss carryforwards that can be utilized annually to offset future taxable income pursuant to IRC Section 382 or similar provisions. Subsequent ownership changes could further affect the limitation in future years. The Company has not completed a study to assess whether a change of control has occurred or whether there have been multiple changes of control since the Company’s formation due to the significant complexity and cost associated with such study and because there could be additional changes in control in the future. As a result, the Company is not able to estimate the effect of the change in control, if any, on the Company’s ability to utilize net operating loss and research and development credit carryforwards in the future.
16. Subsequent Events
On March 7, 2024, the Company’s wholly owned subsidiary Quantum Leap Energy received gross proceeds of $
On April 9, 2024, the Company received approximately $
| 87 |
| Table of Contents |
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
Item 9A. Controls and Procedures
Disclosure Controls and Procedures
Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, evaluated the effectiveness of our disclosure controls and procedures as of December 31, 2023. The term “disclosure controls and procedures,” as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act, mean controls and other procedures of a company that are designed to ensure that information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms. Disclosure controls include, without limitation, controls and procedures designed to ensure that information required to be disclosed by a company on the reports that it files or submits under the Exchange Act is accumulated and communicated to management, including, our principal executive and principal financial officers, as appropriate, to allow timely decisions regarding required disclosure.
Management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving their objectives and management necessarily applies its judgement in evaluating the cost-benefit relationship of possible controls and procedures. Based on the evaluation of our disclosure controls and procedures as of December 31, 2023, our Chief Executive Officer and Chief Financial Officer concluded that, as a result of a material weakness identified in our internal control over financial reporting, our disclosure controls and procedures were not effective as of December 31, 2023.
In the course of preparing the financial statements that are included in this Form 10-K, management has determined that a material weakness exists within the internal controls over financial reporting. The material weakness identified relates to the lack of formal control documentation and consistent execution of control procedures, and the lack of a sufficient complement of personnel within the finance and accounting function with an appropriate degree of knowledge, experience and training. We also noted a material weakness related to logical security and privileged access in the area of information technology. We concluded that the material weaknesses in our internal control over financial reporting information technology occurred because, prior to becoming a public company, we were a private company and did not have the necessary business processes, systems, personnel, and related internal controls necessary to satisfy the accounting and financial reporting requirements of a public company.
In order to remediate the material weaknesses, we expect to enhance our formal documentation over internal control procedures and management controls infrastructure to allow for more consistent execution of control procedures and hire additional accounting, and finance and information technology resources or consultants with public company experience.
We may not be able to fully remediate the identified material weakness until the steps described above have been completed and our internal controls have been operating effectively for a sufficient period of time. We believe we have already and will continue to make progress in our remediation plan during the year ending December 31, 2023, but cannot assure you that we will be able to fully remediate the material weakness in 2024. If the steps we take do not correct the material weakness in a timely manner, we will be unable to conclude that we maintain effective internal control over financial reporting. Accordingly, there could continue to be a reasonable possibility that a material misstatement of our financial statements would not be prevented or detected on a timely basis. We also may incur significant costs to execute various aspects of our remediation plan but cannot provide a reasonable estimate of such costs at this time.
Management’s Annual Report on Internal Controls Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting (as such term is defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act). Our internal control system was designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes, in accordance with generally accepted accounting principles in the United States. Due to inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness of the internal control over financial reporting to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with policies and procedures may deteriorate. Our management, under the supervision and with the participation of our chief executive officer and chief financial officer, evaluated the effectiveness of our internal control over financial reporting as of the end of the period covered by this Annual Report on Form 10-K based on the framework in Internal Control---Integrated Framework (2013 framework) issued by the Committee of Sponsoring Organizations of the Treadway Commission, or COSO. Based on such evaluation, our management concluded that our internal control over financial reporting was effective as of the end of the period covered by this Annual Report on Form 10-K.
| 88 |
| Table of Contents |
This Annual Report on Form 10-K does not include an attestation report on internal control over financial reporting issued by our independent registered public accounting firm. Our auditors will not be required to opine on the effectiveness of our internal control over financial reporting pursuant to Section 404(b) of the Sarbanes-Oxley Act of 2002 until we are no longer an emerging growth company, as defined in the Jumpstart Our Business Startups Act of 2012.
Changes in Internal Control Over Financial Reporting
There were no changes in our internal control over financial reporting that occurred during our most recent fiscal quarter that materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
Item 9B. Other Information
None.
Amended and Restated Bylaws
On April 5, 2024, in connection with certain recent changes to the Delaware General Corporation Law (the “DGCL”) and following the adoption of Rule 14a-19 (the Universal Proxy Rules) under the Securities Exchange Act of 1934, as amended, and a periodic review of the bylaws of ASP Isotopes Inc., the board of directors adopted amended and restated bylaws (the “Amended and Restated Bylaws”), effective immediately. Among other things, the Amended and Restated Bylaws:
| · | enhance the procedural mechanics and disclosure requirements in connection with stockholder nominations of directors and other proposals made in connection with annual and special meetings of stockholders by, among other things: |
| o | requiring additional background information and disclosures regarding proposing stockholders and proposed nominees; |
|
|
|
| o | requiring any stockholder submitting a nomination notice to make a representation as to whether such stockholder intends to solicit proxies in support of director nominees other than the Company’s nominees in accordance with Rule 14a-19 under the Exchange Act and to provide reasonable evidence that certain requirements of such rule have been satisfied; |
|
|
|
| o | clarifying that the Company will disregard the nomination of each proposed director nominee other than the Company’s nominees (notwithstanding that the nominee is included as a nominee in the Company’s proxy statement, notice of meeting or other proxy materials for any annual meeting (or any supplement thereto) and notwithstanding that proxies or votes in respect of the election of such proposed nominees may have been received by the Company (which proxies and votes shall be disregarded)) if, after a stockholder provides notice pursuant to Rule 14a-19 under the Exchange Act, such stockholder subsequently fails to comply with the requirements of Rule 14a-19 under the Exchange Act; |
|
|
|
| o | prohibiting stockholders from nominating placeholder nominees; and |
|
|
|
| o | requiring any stockholder directly or indirectly soliciting proxies from other stockholders to use a proxy card other than white, with the white proxy card being reserved for exclusive use by the board of directors; |
| · | modify the provisions relating to the list of stockholders entitled to vote at stockholder meetings to reflect recent amendments to the DGCL; and |
|
|
|
| · | make other updates, including conforming changes. |
The foregoing summary and description of the Amended and Restated Bylaws does not purport to be complete and is qualified in its entirety by reference to, and should be read in conjunction with, the full text of the Amended and Restated Bylaws, a copy of which is filed as Exhibit 3.2 hereto and is incorporated herein by reference.
Amendments to Employment Agreements
The Chief Executive Officer and Chief Financial Officer are eligible to receive annual bonuses in accordance with the terms of their respective employment agreements, with the amount of such bonus and whether such bonus is paid in cash or stock (or a mix of cash and stock) to be determined by the board of directors in its discretion, based on a recommendation of the compensation committee. On April 5, 2024, in recognition of the contributions of Paul Mann and Robert Ainscow to the company in 2023, the board of directors approved, based on a recommendation by the compensation committee, a discretionary cash bonus for 2023 of $440,000 and $90,000, respectively.
In addition, on April 5, 2024, the board of directors approved amendments to the employment agreements with Messrs. Mann and Ainscow and Dr. Strydom. As a result of the amendments to their respective employment agreements, Mr. Mann was appointed Executive Chairman and Chief Executive Officer and Mr. Ainscow was appointed Chief Operating Officer and Chief Financial Officer; and the base salaries of Mr. Mann, Mr. Ainscow and Dr. Strydom were increased to $520,000, $360,000 and $180,000, respectively.
Item 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections
None.
| 89 |
| Table of Contents |
PART III
Item 10. Directors, Executive Officers and Corporate Governance
The information required by this item will be included in an amendment to this Annual Report on Form 10-K or incorporated by reference from our definitive proxy statement to be filed pursuant to Rule 14A.
Item 11. Executive Compensation
The information required by this item will be included in an amendment to this Annual Report on Form 10-K or incorporated by reference from our definitive proxy statement to be filed pursuant to Rule 14A.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The information required by this item will be included in an amendment to this Annual Report on Form 10-K or incorporated by reference from our definitive proxy statement to be filed pursuant to Rule 14A.
Item 13. Certain Relationships and Related Transactions and Director Independence
The information required by this item will be included in an amendment to this Annual Report on Form 10-K or incorporated by reference from our definitive proxy statement to be filed pursuant to Rule 14A.
Item 14. Principal Accounting Fees and Services
The information required by this item will be included in an amendment to this Annual Report on Form 10-K or incorporated by reference from our definitive proxy statement to be filed pursuant to Rule 14A.
| 90 |
| Table of Contents |
PART IV
Item 15. Exhibits, Financial Statement Schedules
The following documents are filed as part of this Annual Report on Form 10-K:
(a) Financial Statements
The information concerning our consolidated financial statements and Report of Independent Registered Public Accounting Firm required by this Item is incorporated by reference herein to the section of this Annual Report on Form 10-K in Item 8, entitled “Financial Statements and Supplementary Data.”
(b) Financial Statement Schedules
All schedules have been omitted because the required information is not present or not present in amounts sufficient to require submission of the schedules, or because the information required is included in the Financial Statements or notes thereto.
(c) Exhibits
The list of exhibits filed with this report is set forth in the Exhibit Index immediately preceding the signature page and is incorporated herein by reference.
Exhibit |
|
|
Number |
| Description of Document |
| ||
|
|
|
| ||
|
|
|
| Description of Securities Registered Under Section 12 of the Securities Exchange Act of 1934. | |
|
|
|
| ||
|
|
|
| Placement Agent Common Stock Purchase Warrant dated March 17, 2023. | |
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
|
| 91 |
| Table of Contents |
| 92 |
| Table of Contents |
| Release Agreement, dated March 23, 2023 between Revere Securities LLC and ASP Isotopes Inc. | |
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| Consent of EisnerAmper LLP, independent registered public accounting firm. | |
|
|
|
| Power of Attorney (included as part of the signature page to this report). | |
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| Policy Relating to Recovery of Erroneously Awarded Compensation, effective October 2, 2023. | |
|
|
|
| ||
|
|
|
| ||
|
|
|
101.INS |
| Inline XBRL Instance Document – the instance document does not appear in the Interactive Data File because XBRL tags are embedded within the Inline XBRL document. |
|
|
|
101.SCH |
| Inline XBRL Taxonomy Extension Schema Document |
|
|
|
101.CAL |
| Inline XBRL Taxonomy Extension Calculation Linkbase Document |
|
|
|
101.DEF |
| Inline XBRL Taxonomy Extension Definition Linkbase Document |
|
|
|
101.LAB |
| Inline XBRL Taxonomy Extension Label Linkbase Document |
|
|
|
101.PRE |
| Inline XBRL Taxonomy Extension Presentation Linkbase Document |
|
|
|
104 |
| Cover Page Interactive Data File (embedded within the Inline XBRL document) |
____________
* | Filed herewith. |
|
|
** | Furnished herewith. |
|
|
+ | Management contract or compensatory plan or arrangement. |
Item 16. Form 10-K Summary
None.
| 93 |
| Table of Contents |
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized, on the 10th day of April, 2024.
ASP ISOTOPES Inc. |
| ||
|
|
| |
| By | /s/ PAUL E. MANN |
|
|
| Paul E. Mann |
|
|
| Chairman, Chief Executive Officer and Director |
|
We, the undersigned directors and officers of ASP Isotopes Inc., hereby severally constitute Paul E. Mann and Robert Ainscow, and each of them singly, as our true and lawful attorneys with full power to each of them to sign for us, in our names in the capacities indicated below, any and all amendments to this Annual Report on Form 10-K filed with the Securities and Exchange Commission.
This power of attorney may only be revoked by a written document executed by the undersigned that expressly revokes this power by referring to the date and subject hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant in the capacities and on the dates indicated.
Signature |
| Title |
| Date |
|
|
|
|
|
/s/ PAUL E. MANN |
| Chief Executive Officer, Chairman and Director (Principal Executive Officer) |
| April 10, 2024 |
Paul E. Mann |
|
| ||
|
|
|
|
|
/s/ ROBERT AINSCOW |
| Chief Operating Officer and Chief Financial Officer (Principal Financial and Accounting Officer) |
| April 10, 2024 |
Robert Ainscow |
|
| ||
|
|
|
|
|
/s/ MICHAEL GORLEY, Ph.D. |
| Director |
| April 10, 2024 |
Michael Gorley, Ph.D. |
|
| ||
|
|
|
|
|
/s/ DUNCAN MOORE, Ph.D. |
| Director |
| April 10, 2024 |
Duncan Moore, Ph.D. |
|
| ||
|
|
|
|
|
/s/ ROBERT RYAN |
| Director |
| April 10, 2024 |
Robert Ryan |
|
| ||
|
|
|
|
|
/s/ HENDRIK STRYDOM, Ph.D. |
| Director |
| April 10, 2024 |
Hendrik Strydom, Ph.D. |
|
| ||
|
|
|
|
|
/s/ TODD WIDER, M.D. |
| Director |
| April 10, 2024 |
Todd Wider, M.D. |
|
|
| 94 |